Your new post is loading...
 Your new post is loading...
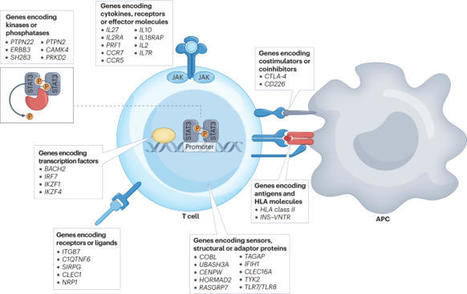
Following the seminal discovery of insulin a century ago, treatment of individuals with type 1 diabetes (T1D) has been largely restricted to efforts to monitor and treat metabolic glucose dysregulation. The recent regulatory approval of the first immunotherapy that targets T cells as a means to delay the autoimmune destruction of pancreatic β-cells highlights the critical role of the immune system in disease pathogenesis and tends to pave the way for other immune-targeted interventions for T1D. Improving the efficacy of such interventions across the natural history of the disease will probably require a more detailed understanding of the immunobiology of T1D, as well as technologies to monitor residual β-cell mass and function. Here we provide an overview of the immune mechanisms that underpin the pathogenesis of T1D, with a particular emphasis on T cells. The first immune-targeted drug for type 1 diabetes (T1D), teplizumab, received regulatory approval by the US FDA in 2022. In this Review, Herold, Walker and colleagues examine the immune mechanisms that underpin T1D and provide an overview of immune-targeted strategies for T1D that are currently in development.
Background Enteroviruses are routinely detected with molecular methods within large cohorts that are at risk of type 1 diabetes. We aimed to examine the association between enteroviruses and either islet autoimmunity or type 1 diabetes. Methods For this systematic review and meta-analysis, we searched PubMed and Embase for controlled observational studies from inception until Jan 1, 2023. Cohort or case-control studies were eligible if enterovirus RNA or protein were detected in individuals with outcomes of islet autoimmunity or type 1 diabetes. Studies in pregnancy or other types of diabetes were excluded. Data extraction and appraisal involved author contact and deduplication, which was done independently by three reviewers. Study quality was assessed with the Newcastle-Ottawa Scale and National Health and Medical Research Council levels of evidence. Pooled and subgroup meta-analyses were done in RevMan version 5.4, with random effects models and Mantel-Haenszel odds ratios (ORs; 95% CIs). The study is registered with PROSPERO, CRD42021278863. Findings The search returned 3266 publications, with 897 full texts screened. Following deduplication, 113 eligible records corresponded to 60 studies (40 type 1 diabetes; nine islet autoimmunity; 11 both), comprising 12077 participants (5981 cases; 6096 controls). Study design and quality varied, generating substantial statistical heterogeneity. Meta-analysis of 56 studies showed associations between enteroviruses and islet autoimmunity (OR 2·1, 95% CI 1·3–3·3; p=0·002; n=18; heterogeneity χ2/df 2·69; p=0·0004; I2=63%), type 1 diabetes (OR 8·0, 95% CI 4·9–13·0; p<0·0001; n=48; χ2/df 6·75; p<0·0001; I2=85%), or within 1 month of type 1 diabetes (OR 16·2, 95% CI 8·6–30·5; p<0·0001; n=28; χ2/df 3·25; p<0·0001; I2=69%). Detection of either multiple or consecutive enteroviruses was associated with islet autoimmunity (OR 2·0, 95% CI 1·0–4·0; p=0·050; n=8). Detection of Enterovirus B was associated with type 1 diabetes (OR 12·7, 95% CI 4·1–39·1; p<0·0001; n=15). Interpretation These findings highlight the association between enteroviruses and islet autoimmunity or type 1 diabetes. Our data strengthen the rationale for vaccine development targeting diabetogenic enterovirus types, particularly those within Enterovirus B. Prospective studies of early life are needed to elucidate the role of enterovirus timing, type, and infection duration on the initiation of islet autoimmunity and the progression to type 1 diabetes. Published in The Lancet (June 27, 2023):
Via Juan Lama
Summary. Type 1 diabetes (T1D) results from autoimmune destruction of insulin-producing beta cells in pancreatic islets. Various immune cell populations are inv
A new treatment using stem cells that produce insulin has surprised experts and given them hope for the 1.5 million Americans living with the disease.
A new study has shown that treating type 1 diabetes-prone mice with the small protein MOTS-c prevented the immune system from destroying insulin-producing pancreatic cells, effectively preventing the onset of the autoimmune disease.
Understanding the pathogenesis of type 1 diabetes (T1D), in order to direct therapy for prevention and treatment is a major goal of research into T1D. At the International Congress of the Immunolog
Sci Transl Med. 2017 Feb 22;9(378). pii: eaaf8848. doi: 10.1126/scitranslmed.aaf8848.
Susceptibility to type 1 diabetes involves genetic risk factors and potential environmental triggers, making it harder to predict which individuals will actually develop disease. Longitudinal samples collected starting shortly after birth from children at risk for type 1 diabetes were examined by Heninger et al . to scrutinize early immune responses. Some, but not all, of these children eventually developed autoimmunity; CD4+ T cells from those that progressed had a distinct phenotype after presentation of autoantigen, suggesting that they had already been somewhat activated, even before autoantibodies were detectable. These exciting findings indicate that cues that dictate type 1 diabetes development may be happening even earlier than expected, in infancy.
Abby L. Dotson, Lesya Novikova, Lisa Stehno-Bittel, Stephen H. Benedict
Clinical Immunology, Volume 148, Issue 2, August 2013, Pages...
|
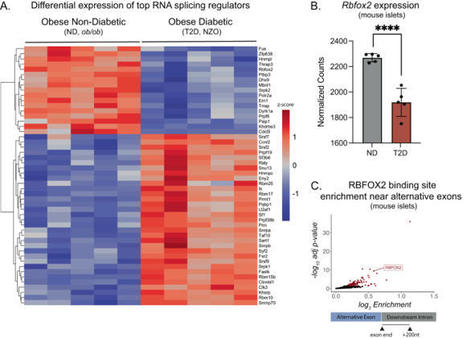
Insulin secretion from the pancreatic beta cell is a tightly regulated process that is vital for maintaining blood glucose homeostasis. Here, the authors show that the RNA binding protein RBFOX2 is a regulator of insulin secretion through the alternative splicing of genes required for insulin...

|
Suggested by
LIGHTING
|
Abstract Emerging evidence indicates a bi-directional relationship between SARS-CoV-2 and diabetes. The possibility exists that SARS-CoV-2 could induce diabetes, but it is not yet clear whether thi...
Commemorating 100 years since the discovery of insulin The discovery of insulin 100 years ago heralded a dramatic shift in the treatment of type 1 diabetes (T1D). Until that point, T1D had been a fatal disease with no treatment options, but the discovery of insulin transformed T1D into a chronic disease that could be managed by insulin therapy.
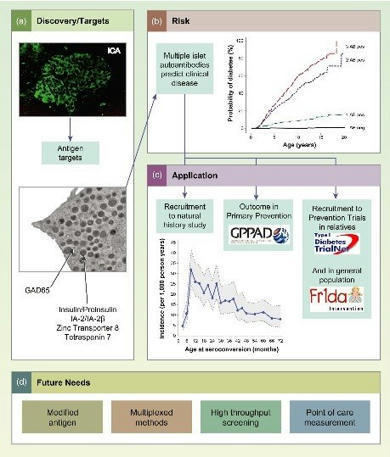
The role of autoantibodies in the early diagnosis of type-1 diabetes. The discovery of a new CD4 T cell autoantigenic target that facilitates this. The mechanisms by which immune cells subsequently infiltrate the pancreas.
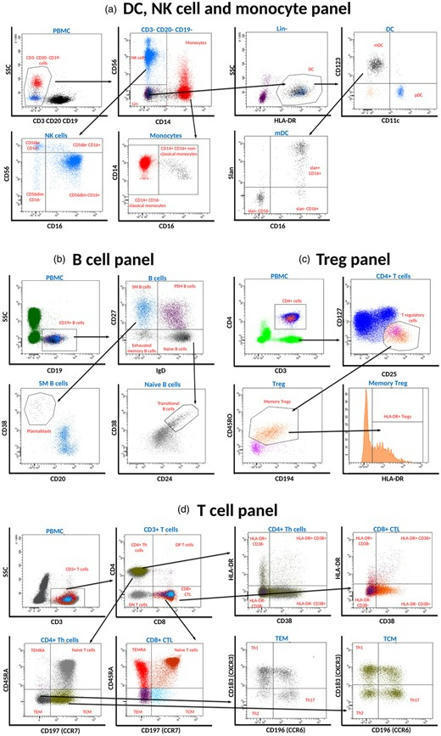
The phenotype of autoreactive T cells in type 1 diabetes is described as Th1, Th17 and/or Th21, but is largely uncharacterized.We combined multi-para…...
Although natural killer (NK) cells have been implicated in the pathogenesis of type 1 diabetes, the mechanism whereby they contribute to this autoimmune disorder—which depends on destruction of pancreatic beta cells by autoreactive T cells—has been unclear. Gur et al . wondered whether natural cytotoxicity receptor NKp46, an activating NK cell receptor that has been implicated in tumor immunity and control of influenza despite having no known ligand (see Colucci and Cilio), might play a role. They found that fusion proteins of NKp46 or the mouse ortholog NCR1 with immunoglobulin (NKp46-Ig and NCR1-Ig, respectively) recognized human and mouse beta cells but no other pancreatic cells. Immunohistochemical analysis of nonobese diabetic mice (NOD mice, a model of type 1 diabetes) revealed that the NKp46 ligand was present before the development of the pancreatic islet inflammation that precedes diabetes (insulinitis) and throughout the development of diabetes. A comparison of mouse NK cells with NCR1 to NK cells lacking NCR1 indicated that, although beta cells failed to elicit cytokine secretion, they stimulated NK cell degranulation; indeed, NK cells isolated from islets of prediabetic NOD mice showed substantial degranulation. Mice lacking NCR1 were resistant to the development of type 1 diabetes in response to injection with low-dose streptozotocin (LDST), and NK cells appeared in the pancreas of NOD and LDST mice during insulinitis, becoming most abundant at the prediabetic stage, during the transition from insulinitis to diabetes. Injection of NKp46-Ig or NCR1-Ig into NOD mice, which stimulated the production of antibodies directed against NKp46 or NCR1, respectively, decreased the surface abundance of NCR1, inhibited beta cell–induced NK cell degranulation, and prevented the development of diabetes, even when administered at the late prediabetic stage. The authors thus conclude that a ligand to NKp46 is present on pancreatic beta cells and that this NK cell receptor is crucial to the development of type 1 diabetes.
C. Gur, A. Porgador, M. Elboim, R. Gazit, S. Mizrahi, N. Stern-Ginossar, H. Achdout, H. Ghadially, Y. Dor, T. Nir, V. Doviner, O. Hershkovitz, M. Mendelson, Y. Naparstek, O. Mandelboim, The activating receptor NKp46 is essential for the development of type 1 diabetes. Nat. Immunol. 11 , 121–128 (2010). [[PubMed]][1]
F. Colucci, C. M. Cilio, Taming killer cells may halt diabetes progression. Nat. Immunol. 11 , 111–112 (2010). [[PubMed]][2]
[1]: http://www.ncbi.nlm.nih.gov/pubmed/20023661
[2]: http://www.ncbi.nlm.nih.gov/pubmed/20084067
N-terminally truncated (96–585) GAD65 (tGAD65) autoantibodies may better delineate type 1 diabetes than full-length GAD65 (fGAD65) autoantibodies. We aimed to compare the diagnostic sensitivity and specificity between fGAD65 and tGAD65 autoantibodies for type 1 diabetes in relation to HLA-DQ. Sera from children and adolescents with newly diagnosed type 1 diabetes ( n = 654) and healthy control subjects ( n = 605) were analyzed in radiobinding assays for fGAD65 (fGADA), tGAD65 (tGADA), and commercial 125I-GAD65 (RSRGADA) autoantibodies. The diagnostic sensitivity and specificity in the receiver operating characteristic curve did not differ between fGADA and tGADA. At the optimal cutoff, the diagnostic sensitivity for fGADA was lower than tGADA at similar diagnostic specificities. In 619 patients, 64% were positive for RSRGADA compared with 68% for fGADA and 74% for tGADA. Using non-DQ2/non-DQ8 patients as reference, the risk of being diagnosed with fGADA and tGADA was increased in patients with DQ2/2 and DQ2/8. Notably, logistic regression analysis suggested that DQ8/8 patients had an increased risk to be diagnosed with tGADA ( P = 0.003) compared with fGADA ( P = 0.09). tGADA had a higher diagnostic sensitivity for type 1 diabetes than both fGADA and RSRGADA. As DQ8/8 patients represent 10–11% of patients with newly diagnosed type 1 diabetes <18 years of age, tGADA analysis should prove useful for disease classification.
Immunotherapy. 2014 Dec;6(12):1239-42. doi: 10.2217/imt.14.96.
|

 Your new post is loading...
Your new post is loading...
 Your new post is loading...
Your new post is loading...