Cancer immunotherapy utilizing Vγ9Vδ2 T cells has been developed over the past decade. A large number of clinical trials have been conducted on various types of solid tumors as well as hematological malignancies. Vγ9Vδ2 T cell-based immunotherapy can be classified into two categories based on the methods of activation and expansion of these cells. Although the in vivo expansion of Vγ9Vδ2 T cells by phosphoantigens or nitrogen-containing bisphosphonates (N-bis) has been translated to early-phase clinical trials, in which the safety of the treatment was confirmed, problems such as activation-induced Vγ9Vδ2 T cell anergy and a decrease in the number of peripheral blood Vγ9Vδ2 T cells after infusion of these stimulants have not yet been solved. In addition, it is difficult to ex vivo expand Vγ9Vδ2 T cells from advanced cancer patients with decreased initial numbers of peripheral blood Vγ9Vδ2 T cells. In this article, we review the clinical studies and reports targeting Vγ9Vδ2 T cells and discuss the development and improvement of Vγ9Vδ2 T cell-based cancer immunotherapy.
Via Krishan Maggon



 Your new post is loading...
Your new post is loading...







Pharmaceuticals 2015, 8(1), 40-61; doi:10.3390/ph8010040
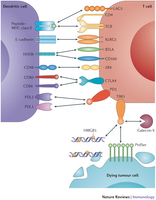
Reviewγδ T Cell Immunotherapy—A ReviewHirohito Kobayashi 1,* and Yoshimasa Tanaka 2,*1Transfusion Medicine and Cell Processing, Department of Urology, Tokyo Women’s Medical University, 8-1 Kawada-cho, Shinjuku-ku, Tokyo 162-8666, Japan2Center for Therapeutic Innovation, Graduate School of Biomedical Sciences, Nagasaki University, 1-14 Bunkyo-machi, Nagasaki 852-8521, Japan*Authors to whom correspondence should be addressed; E-Mails: hirohitokobayashi-jua@umin.ac.jp (H.K.); ystanaka@nagasaki-u.ac.jp (Y.T.); Tel.: +81-3-3353-8111 (ext. 25035) (H.K.); +81-95-819-2890 (Y.T.); Fax: +81-3-5269-7685 (H.K.); +81-95-819-2420 (Y.T.).Academic Editor: Shin MineishiReceived: 6 January 2015 / Accepted: 2 February 2015 / Published: 12 February 2015- See more at: http://www.mdpi.com/1424-8247/8/1/40/htm#sthash.jOThc0Xd.dpuf