 Your new post is loading...
 Your new post is loading...
Abstract Lung cancer remains the most common cause of cancer-related deaths in the United States, with SEER data showing lung cancer accounting for 29% of all male-related cancer mortality and 26% of all female-related mortality. Patients with small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC) who have localized disease both have 5-year survival rates of 52.2%, whereas patients with metastatic disease have 5-year survival rates of only 3.7%. Traditional anti-cancer therapies (surgery, radiotherapy, and chemotherapy) have limited effectiveness in curbing progression. However, advances in immunology and molecular biology in the past two decades have resulted in improved prognosis for those with SCLC and NSCLC, although novel therapies are still needed to make significant improvements in median overall and progression-free survival rates. Notable progress on the importance of tumor immunology has included work on immune surveillance, antigenic targets, and immune checkpoints. Immunotherapies, including vaccines, which can induce antitumor responses by harnessing the power of the immune system, may help to fill this void, and the cancer vaccine continues to be studied as adjunctive therapy. Here, we review recently reported results from clinical trials as well as the possible future roles of vaccine therapy in the treatment of SCLC and NSCLC patients.
Via Krishan Maggon
There are many reasons to be excited by the science being presented at this year’s American Society for Clinical Oncology meeting. The data...
Via Krishan Maggon
Highlights • CD154 is a co-stimulatory molecule known for its role in inflammatory and autoimmune diseases. • CD154 and its receptor, CD40 are expressed in many types of tumors. • The CD154/CD40 interaction is capable of inducing the proliferation and rescue from apoptosis of tumor cells. • CD154 activates anti-tumoral immunity, and by engaging CD40 may induce apoptosis of tumor cells. • The role of CD154 in cancer immunotherapy was demonstrated in animal models and clinically assessed in patients.
Via Krishan Maggon
Abstract Cancer vaccines are designed to promote tumor specific immune responses, particularly cytotoxic CD8 positive T cells that are specific to tumor antigens. The earliest vaccines, which were developed in 1994-95, tested non-mutated, shared tumor associated antigens that had been shown to be immunogenic and capable of inducing clinical responses in a minority of people with late stage cancer. Technological developments in the past few years have enabled the investigation of vaccines that target mutated antigens that are patient specific. Several platforms for cancer vaccination are being tested, including peptides, proteins, antigen presenting cells, tumor cells, and viral vectors. Standard of care treatments, such as surgery and ablation, chemotherapy, and radiotherapy, can also induce antitumor immunity, thereby having cancer vaccine effects. The monitoring of patients’ immune responses at baseline and after standard of care treatment is shedding light on immune biomarkers. Combination therapies are being tested in clinical trials and are likely to be the best approach to improving patient outcomes.
Via Krishan Maggon
An experimental antibody significantly reduced HIV levels in infected people for as long as 28 days. The approach might help to combat a wide range of HIV strains. In the current study, the researchers evaluated one of these promising monoclonal anti-HIV antibodies in people. The small phase 1 clinical trial included 29 volunteers—17 HIV-infected and 12 uninfected. The study was supported by the Bill and Melinda Gates Foundation, NIH’s National Institute of Allergy and Infectious Diseases (NIAID), and others. Results appeared online on April 8, 2015, in Nature. The participants received a single intravenous dose (of 1, 3, 10, or 30 milligrams per kilogram of body weight) of the experimental antibody called 3BNC117. The antibody was well-tolerated by all participants. Among those infected with HIV, the 2 lower doses caused small and transient changes in blood HIV levels. The 8 participants who received the highest dose, however, had significant and rapid decreases in HIV. The virus’s resistance to the antibody was variable. In some people, HIV remained sensitive to 3BNC117 for 28 days.
Via Krishan Maggon
The MaxCyte GT® Flow Transfection System is a universal platform technology for rapid, automated loading of CAR (chimeric antigen receptor) mRNA into peripheral blood mononuclear cells using closed-system aseptic processing.
Via Krishan Maggon
Tumor-specific immunotherapy holds the promise of eradicating malignant tumors with exquisite precision without additional toxicity to standard treatments.
Via Krishan Maggon
Monoclonal antibodies (mAbs) have inaugurated the concepts of tumor-targeted therapy and personalized medicine. A new family of mAbs is currently emerging in the clinic, which target immune cells rather than cancer cells. These immune-targeted therapies have recently demonstrated long-term tumor responses in adults with refractory/relapsing metastatic solid tumors. Pediatric cancers are different from their adult counterparts in terms of histological features and immune infiltrates. However, the same immune checkpoint targets can be expressed within the microenvironment of pediatric tumors. The benefits of immune checkpoint blockade in pediatric cancers are currently under evaluation in early phase clinical trials. Pediatr Blood Cancer © 2015 Wiley Periodicals, Inc.
Via Krishan Maggon
(2015). Macrophages are critical effectors of antibody therapies for cancer. mAbs: Vol. 7, No. 2, pp. 303-310.
Via Krishan Maggon
Abstract The recent successes of adoptive T-cell immunotherapy for the treatment of hematologic malignancies have highlighted the need for manufacturing processes that are robust and scalable for product commercialization. Here we review some of the more outstanding issues surrounding commercial scale manufacturing of personalized-adoptive T-cell medicinal products. These include closed system operations, improving process robustness and simplifying work flows, reducing labor intensity by implementing process automation, scalability and cost, as well as appropriate testing and tracking of products, all while maintaining strict adherence to Current Good Manufacturing Practices and regulatory guidelines. A decentralized manufacturing model is proposed, where in the future patients’ cells could be processed at the point-of-care in the hospital.
Via Krishan Maggon
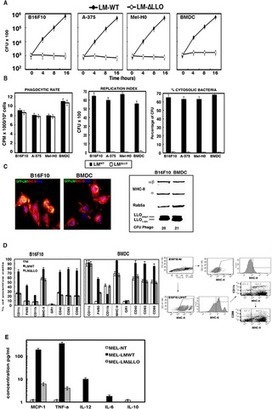
Listeria monocytogenes is a gram-positive bacteria and human pathogen widely used in cancer immunotherapy because of its capacity to induce a specific cytotoxic T cell response in tumours.
Via Krishan Maggon
Abstract The concept of using immunotherapy to treat melanoma has existed for decades. The rationale comes from the knowledge that many patients with melanoma have endogenous immune responses against their tumor cells and clinically meaningful tumor regression can be achieved in a minority of patients using cytokines such as interleukin-2 and adoptive cellular therapy. In the last 5 years there has been a revolution in the clinical management of melanoma in large measure based on the development of antibodies that influence T cell regulatory pathways by overcoming checkpoint inhibition and providing co-stimulation, either of which results in significantly more effective immune-mediated tumor destruction. This review will describe the pre-clinical and clinical application of antagonistic antibodies targeting the T-cell checkpoints cytotoxic T-lymphocyte antigen 4 (CTLA-4) and programmed death 1 (PD-1), and agonistic antibodies targeting the costimulatory pathways OX40 and 4-1BB. Recent progress and opportunities for future investigation of combination antibody therapy will be described.
Via Krishan Maggon
Abstract In recent years, immune checkpoints that maintain physiologic self-tolerance have been implicated in the down-regulation of anti-tumor immunity. Efforts to restore latent anti-tumor immunity have focused on antibody-based interventions targeting CTL antigen 4 (CTLA-4) and programmed cell death protein 1 (PD-1) on T lymphocytes and its principal ligand (PD-L1) on tumor cells. Ipilimumab, an antibody targeting CTLA-4, appears to restore tumor immunity at the priming phase, whereas anti-PD-1/PD-L1 antibodies restore immune function in the tumor microenvironment. Although ipilimumab can produce durable long-term responses in patients with advanced melanoma, it is associated with significant immune-related toxicities. By contrast, antibodies targeting either PD-1 or PD-L1 have produced significant anti-tumor activity with considerably less toxicity. Activity was seen in patients with melanoma and renal cancer, as well as those with non-small-cell lung, bladder and head and neck cancers, tumors not previously felt to be sensitive to immunotherapy. The tolerability of PD-1-pathway blockers and their unique mechanism of action have made them ideal backbones for combination regimen development. Combination approaches involving cytotoxic chemotherapy, anti-angiogenic agents, alternative immune-checkpoint inhibitors, immunostimulatory cytokines and cancer vaccines are currently under clinical investigation. Current efforts focus on registration trials of single agents and combinations in various diseases and disease settings and identifying predictive biomarkers of response.
Via Krishan Maggon
|
Abstract Scientific insights into the human immune system have recently led to unprecedented breakthroughs in immunotherapy. In the twenty-first century, drugs and cell-based therapies developed to bolster humoral and T cell immunity represent an established and growing component of cancer therapeutics. Although natural killer (NK) cells have long been known to have advantages over T cells in terms of their capacity to induce antigen-independent host immune responses against malignancies, their therapeutic potential in the clinic has been largely unexplored. A growing number of scientific discoveries into pathways that both activate and suppress NK cell function, as well as methods to sensitize tumours to NK cell cytotoxicity, have led to the development of numerous pharmacological and genetic methods to enhance NK cell antitumour immunity. These findings, as well as advances in our ability to expand NK cells ex vivoand manipulate their capacity to home to the tumour, have now provided investigators with a variety of new methods and strategies to harness the full potential of NK cell-based cancer immunotherapy in the clinic.
Via Krishan Maggon
Findings The anti-PD-1 and anti-PD-L1 agents have been reported to have impressive antitumor effects in several malignancies, including melanoma. The greatest clinical activity in unselected patients has been seen in melanoma. Tumor expression of PD-L1 is a suggestive, but inadequate, biomarker predictive of response to immune-checkpoint blockade. However, tumors expressing little or no PD-L1 are less likely to respond to PD-1 pathway blockade. Combination checkpoint blockade with PD-1 plus cytotoxic T-lymphocyte antigen (CTLA)-4 blockade appears to improve response rates in patients who are less likely to respond to single-checkpoint blockade. Toxicity with PD-1 blocking agents is less than the toxicity with previous immunotherapies (eg, interleukin 2, CTLA-4 blockade). Certain adverse events can be severe and potentially life threatening, but most can be prevented or reversed with close monitoring and appropriate management. Implications This family of immune-checkpoint inhibitors benefits not only patients with metastatic melanoma but also those with historically less responsive tumor types. Although a subset of patients responds to single-agent blockade, the initial trial of checkpoint-inhibitor combinations has reported a potential to improve response rates. Combination therapies appear to be a means of increasing response rates, albeit with increased immune-related adverse events. As these treatments become available to patients, education regarding the recognition and management of immune-related effects of immune-checkpoint blockade will be essential for maximizing clinical benefit.
Via Krishan Maggon
(Reuters) - Worldwide spending on cancer medicines reached $100 billion in 2014, an increase of 10.3 percent from 2013 and up from $75 billion five years earlier, according to IMS Health's Global Oncology...
Via Krishan Maggon
This review discusses the development of targeted and immune therapies for advanced melanoma, reviews current patient management, and highlights future directions.
Via Krishan Maggon
At the annual meeting of the American Association of Cancer Researchers, presenters discuss strategies to improve the safety and effectiveness of reengineered T cells in eradicating tumors.
Via Krishan Maggon
"OS & Long-Term Safety of #Nivolumab in Patients With Previously Treated Advanced #NSCLC" JCO http://t.co/TjxJ8tP1Sr Purpose Programmed death 1 is an immune checkpoint that suppresses antitumor immunity. Nivolumab, a fully human immunoglobulin G4 programmed death 1 immune checkpoint inhibitor antibody, was active and generally well tolerated in patients with advanced solid tumors treated in a phase I trial with expansion cohorts. We report overall survival (OS), response durability, and long-term safety in patients with non–small-cell lung cancer (NSCLC) receiving nivolumab in this trial. Patients and Methods Patients (N = 129) with heavily pretreated advanced NSCLC received nivolumab 1, 3, or 10 mg/kg intravenously once every 2 weeks in 8-week cycles for up to 96 weeks. Tumor burden was assessed by RECIST (version 1.0) after each cycle. Results Median OS across doses was 9.9 months; 1-, 2-, and 3-year OS rates were 42%, 24%, and 18%, respectively, across doses and 56%, 42%, and 27%, respectively, at the 3-mg/kg dose (n = 37) chosen for further clinical development. Among 22 patients (17%) with objective responses, estimated median response duration was 17.0 months. An additional six patients (5%) had unconventional immune-pattern responses. Response rates were similar in squamous and nonsquamous NSCLC. Eighteen responding patients discontinued nivolumab for reasons other than progressive disease; nine (50%) of those had responses lasting > 9 months after their last dose. Grade 3 to 4 treatment-related adverse events occurred in 14% of patients. Three treatment-related deaths (2% of patients) occurred, each associated with pneumonitis. Conclusion Nivolumab monotherapy produced durable responses and encouraging survival rates in patients with heavily pretreated NSCLC. Randomized clinical trials with nivolumab in advanced NSCLC are ongoing.
Via Krishan Maggon
The promising clinical results obtained with engineered T cells, including chimeric antigen receptor (CAR) therapy, call for further advancements to facilitate and broaden their applicability. One potentially beneficial innovation is to exploit new T cell sources that reduce the need for autologous cell manufacturing and enable cell transfer across histocompatibility barriers. Here we review emerging T cell engineering approaches that utilize alternative T cell sources, which include virus-specific or T cell receptor-less allogeneic T cells, expanded lymphoid progenitors, and induced pluripotent stem cell (iPSC)-derived T lymphocytes. The latter offer the prospect for true off-the-shelf, genetically enhanced, histocompatible cell therapy products.
Via Krishan Maggon
Abstract
The Society for Immunotherapy of Cancer (SITC) has regularly hosted workshops and working groups focused on immunologic monitoring and immune biomarkers. Due to advances in cancer immunotherapy, including positive results from clinical trials testing new agents and combinations, emerging new technologies for measuring aspects of immunity, and novel candidate biomarkers from early phase trials, the SITC Immune Biomarkers Taskforce has reconvened to review the state of the art, identify current hurdles to further success and to make recommendations to the field. Topics being addressed by individual working groups include: (1) validation of candidate biomarkers, (2) identification of the most promising technologies, (3) testing of high throughput immune signatures and (4) investigation of the pre-treatment tumor microenvironment. Resultant recommendations will be published in JITC.
Via Krishan Maggon
Highlights • T cells undergo metabolic remodeling to support their function. • Metabolic pathways impact on T cell differentiation decisions and function in the periphery. • Manipulating metabolic microenvironments may enhance T cell function in cancer. • Metabolic pathways could be targeted for the treatment of human disease. In the past several years a wealth of evidence has emerged illustrating how metabolism supports many aspects of T cell biology, as well as how metabolic changes drive T cell differentiation and fate. We outline developing principles in the regulation of T cell metabolism, and discuss how these processes are affected in settings of inflammation and cancer. In this context we discuss how metabolic pathways might be manipulated for the treatment of human disease, including how metabolism may be targeted to prevent T cell dysfunction in inhospitable microenvironments, to generate more effective adoptive cellular immunotherapies in cancer, and to direct T cell differentiation and function towards non-pathogenic phenotypes in settings of autoimmunity.
Via Krishan Maggon
RT @CeriFielding: Natural killer cells #Immunotherapy @NatureMedicine http://t.co/R37PGlyaLj" ; Cancer immunotherapy—which trains the body's own immune system to fight tumors—has made medical headlines in the last few years, with analysts projecting that it could give rise to treatments worth $35 billion a year over the next decade. For the most part, cancer immunotherapy has relied on the power of T cells. Now, another class of immune cells—known as natural killer (NK) cells—that can function to kill cancer is nearing its big break. NK cells may hold the potential to kill off cancer cells without damaging healthy tissues or risking the T cell–driven inflammatory cytokine storm that can accompany other immunotherapies. But a complicated assortment of protein receptors that control their function makes NK cells unwieldy and unpredictable. Dr. Porter, UPenn spoke Sunday at the 35th Annual Conference on Clinical Hematology & Oncology, held in La Jolla by Scripps Health.
Via Krishan Maggon
Abstract Immunotherapy for the treatment of cancer is rapidly evolving from therapies that globally and non-specifically simulate the immune system to more targeted activation of individual components of the immune system. The net result of this targeted approach is decreased toxicity and increased efficacy of immunotherapy. More specifically, therapies that inhibit the interaction between programmed death ligand 1 (PD-L1), present on the surface of tumor or antigen-presenting cells, and programmed death 1 (PD-1), present on the surface of activated lymphocytes, are generating much excitement and enthusiasm, even in malignancies that are not traditionally considered to be immunogenic. Herein, we review the current landscape of anti-PD-1 and anti-PD-L1 therapies in the world of oncology. We have performed a comprehensive literature search on the data available through PubMed, Medline, Scopus, the ClinicalTrials.gov registry, and abstracts from major oncology meetings in order to summarize the clinical data of anti-PD-1/PD-L1 therapies.
Via Krishan Maggon
MT @NatRevClinOncol A shed NKG2D ligand that promotes NK cell activation & #tumor rejection http://t.co/fjxcQRyDGS #immunotherapy #cancer Abstract Immune cells, including natural killer (NK) cells, recognize transformed cells and eliminate them in a process termed immunosurveillance. It is thought that tumor cells evade immunosurveillance by shedding membrane ligands that bind to the NKG2D activating receptor on NK cells and/or T cells, and desensitize these cells. In contrast, we show that in mice, shedding of MULT1, a high affinity NKG2D ligand, causes NK cell activation and tumor rejection. Recombinant soluble MULT1 stimulated tumor rejection in mice. Soluble MULT1 functions, at least in part, by competitively reversing a global desensitization of NK cells imposed by engagement of membrane NKG2D ligands on tumor-associated cells, such as myeloid cells. The results overturn conventional wisdom that soluble ligands are inhibitory, and suggest a new approach for cancer immunotherapy.
Via Krishan Maggon
|



 Your new post is loading...
Your new post is loading...



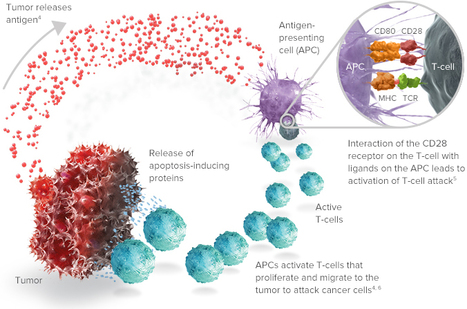
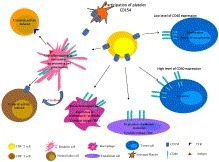








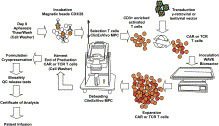
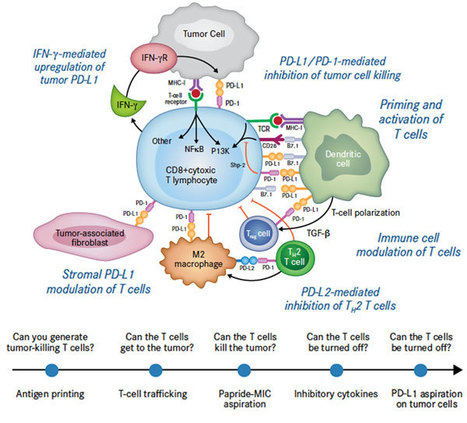

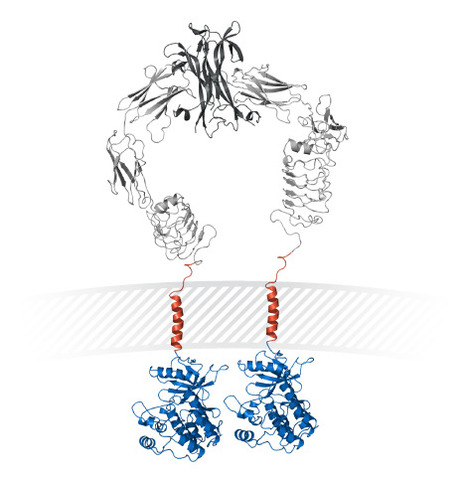





Volume 153, September 2015, Pages 1–9
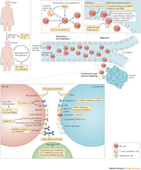
Vaccine immunotherapy in lung cancer: Clinical experience and future directionsMorganna Freeman-Kellera, Jamie Goldmana, Jhanelle Grayb, , doi:10.1016/j.pharmthera.2015.05.004