Your new post is loading...
 Your new post is loading...
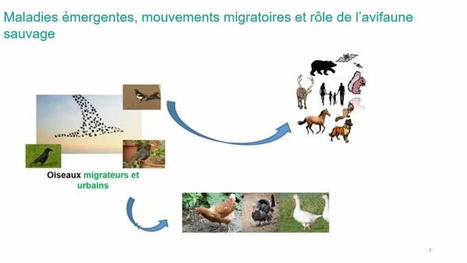
Les présentations du "Webinaire vaccin anti grippe" One Health organisé par le DIM One Health 2.0 et le club de vaccinologie de la SFI sont désormais…
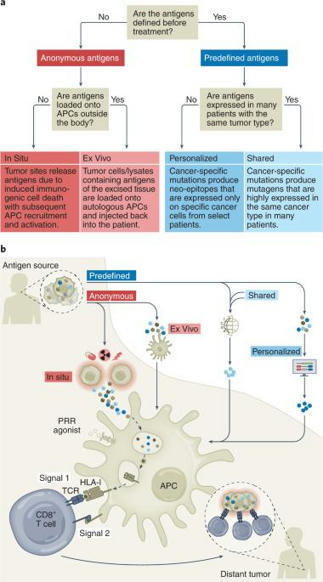
💥 How do we measure cancer vaccine immune responses? Is this similar to (simply..) measuring antibody titers, as in prophylactic vaccines? It took some time…
Abstract. Human CD56br natural killer (NK) cells represent a small subset of CD56+ NK cells in circulation and are largely tissue-resident. The frequency and nu
Malaria is thought to be responsible for up to five percent of all human deaths in the 20th Century.
The world now has two vaccines which, alongside other…
Since the FDA approved the first CAR-T therapy back in August 2017, high prices, small patients pools and limited manufacturing capacity have at times hindered these cell-based treatments. As biopharma companies clear those hurdles, a larger, more systemic problem now threatens the drug class.
Researchers engineered IgA antibodies from IgG monoclonals, enhancing neutralization against Omicron by up to 75-fold and offering protection in mice models.
Checkpoint blockade immunotherapy has marked a turning point in the fight against cancer. It has not only transformed scientific thinking of how our bodies can respond to cancer but has also shown us that it can be possible to tackle cancers we previously viewed as untreatable.
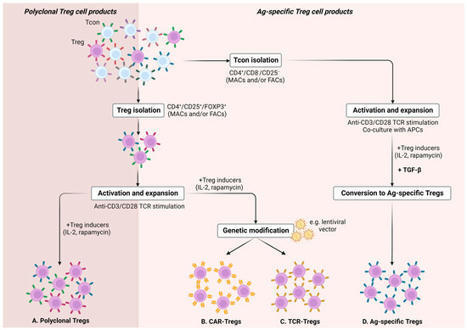
Regulatory T cells (Tregs) are fundamental to maintaining immune homeostasis by inhibiting immune responses to self-antigens and preventing the excessive activation of the immune system. Their functions extend beyond immune surveillance and subpopulations of tissue-resident Treg cells can also...
NIH and New Zealand researchers show how conditioning the lungs with interferon-gamma, a natural immune system protein best known for fighting bacterial…
The development of therapeutic approaches for the induction of robust, long-lasting and antigen-specific immune tolerance remains an important unmet clinical need for the management of autoimmunity, allergy, organ transplantation and gene therapy. Recent breakthroughs in our understanding of immune tolerance mechanisms have opened new research avenues and therapeutic opportunities in this area. Here, we review mechanisms of immune tolerance and novel methods for its therapeutic induction. The induction of antigen-specific immune tolerance is considered the 'holy grail' of disease management for autoimmunity and organ transplantation. Are we getting any closer to this goal? Here, the authors update us on the current progress and challenges to the therapeutic induction of antigen-specific immune tolerance.
Vaccines Europe conducted a pipeline review of its 15 member companies, including information up to August 2023. The review shows a promising vaccines pipeline aimed at tackling the challenges of today and tomorrow, such as the burden of respiratory tract infections, AMR, the ageing of the population, climate change and zoonotic infections, through leveraging a […]
Check out our new Review in European Journal of Immunology Wiley on the past, present and future of rational polymer-based vaccine #adjuvant design! Myself…
|
Repair of lung injury due to viral infection can be facilitated by nanoparticle-delivered mRNA targeting TGF-βR2 signaling. Respiratory infections, such as those caused by SARS-CoV-2 or influenza, can damage the lungs’ delicate network of capillary blood vessels, compromising oxygen delivery and carbon dioxide removal. To overcome this damage, the lungs depend on the regenerative capacities of vascular endothelial cells. As valuable as these cells are, they can, according to University of Pennsylvania scientists, benefit from a little help. The scientists, led by Andrew Vaughan, PhD, focused on a repair pathway involving vascular endothelial growth factor α (Vegfa) and the TGF-β receptor 2 (TGF-βR2). Using animal models and human tissue samples, the scientists showed that delivering Vegfa via lipid nanoparticles (LNPs) greatly enhances modes of repair for damaged blood vessels. “Mice deficient in endothelial Tgfbr2 exhibited prolonged injury and diminished vascular repair,” the article’s authors wrote. “Loss of endothelial Tgfbr2 prevented autocrine Vegfa expression, reduced endothelial proliferation, and impaired renewal of aerocytes thought to be critical for alveolar gas exchange.” “We developed a lipid nanoparticle that targets the pulmonary endothelium, Lung-LNP (LuLNP),” the authors continued. “Delivery of Vegfa mRNA, a critical TGF-βR2 downstream effector, by LuLNPs improved the impaired regeneration phenotype of endothelial cell Tgfbr2 deficiency during influenza injury.” Vaughan’s team and other investigators had previously shown that endothelial cells are among the unsung heroes in repairing the lungs after viral infections. But Vaughan’s team noted that its work demonstrated that a “more granular understanding of the fundamental mechanisms driving reconstitution of lung endothelium” could inform efforts to facilitate therapeutic vascular repair. “Here we’ve identified and isolated pathways involved in repairing this tissue, delivered mRNA to endothelial cells, and consequently observed enhanced recovery of the damaged tissue,” Vaughan said. “These findings hint at a more efficient way to promote lung recovery after diseases like COVID-19.” The team found Vegfa’s involvement in this recovery, while building on work in which they used single-cell RNA sequencing to identify TGF-βR2 as a major signaling pathway. The researchers saw that when TGF-βR2 was missing, it stopped the activation of Vegfa. This lack of signal made the blood vessel cells less able to multiply and renew themselves, which is vital for the exchange of oxygen and carbon dioxide in the tiny air sacs of the lungs. “We’d known there was a link between these two pathways, but this motivated us to see if delivering Vegfa mRNA into endothelial cells could improve lung recovery after disease-related injury,” said first author Gan Zhao, PhD, a postdoctoral researcher in the Vaughan laboratory. The Vaughan laboratory then reached out to Michael J. Mitchell, PhD, of the School of Engineering and Applied Science, whose laboratory specializes in LNPs, to see if delivery of this mRNA cargo would be feasible. “LNPs have been great for vaccine delivery and have proven incredibly effective delivery vehicles for genetic information,” said Mitchell, who is an associate professor of bioengineering at Penn Engineering and a co-author of the paper. “But the challenge here was to get the LNPs into the bloodstream without them heading to the liver, which is where they tend to congregate as its porous structure lends favor to substances passing from the blood into hepatic cells for filtration. So, we had to devise a way to specifically target the endothelial cells in the lungs.” The Mitchell laboratory’s LNPs proved effective in delivering Vegfa into endothelial cells, and as a result, the researchers saw a marked improvement in vascular recovery in their animal models. Within the animal models, the researchers saw improved oxygen levels, and in some, the treatment helped them recover their weight better than the control group. These treated mice also had less lung inflammation, shown by lower levels of certain markers in their lung fluid, and their lungs showed less damage and scarring, with more healthy blood vessels. “We’re looking forward to testing this delivery platform for other cell types in the lung, and it will be important to evaluate whether TGF-βR2 signaling is important in other injury contexts including chronic conditions like emphysema and chronic obstructive pulmonary disease,” Vaughan said. “With this proof-of-concept being well validated, we’re sure that we’ll pave the way for new mRNA-based strategies for treating lung injury.” Published in January 31, 2024 in Science Translational Med: https://doi.org/10.1126/scitranslmed.adg6229
Via Juan Lama
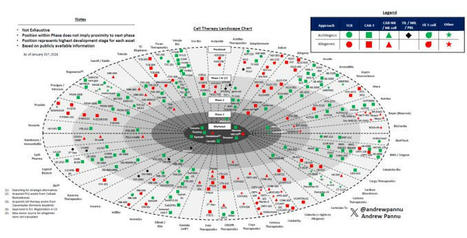
Is the best yet to come for cell therapy?
I pulled together 45 companies in the space and charted the preclinical and clinical assets of each, segmented by… | 47 commentaires sur LinkedIn
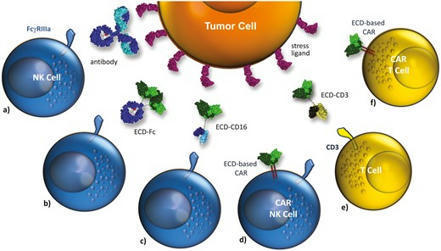
Natural Killer (NK) cells exert an important role in cancer immune surveillance and recognition of malignant cells as well as their controlled activation is fac
Real-world data suggests the HPV vaccine dramatically cuts cancer risk among women who received it during adolescence.
The success of CAR-T cell therapies is dependent on effective cell manufacturing that impacts product safety, efficacy, and patient accessibility. Here, we disc
Human Vγ9Vδ2 T lymphocytes are regarded as promising effector cells for cancer immunotherapy since they have the ability to eliminate several tumor cells through non-peptide antigen recognition. However, the cytotoxic function and the mechanism of Vγ9Vδ2 T cells leading to specific killing of...
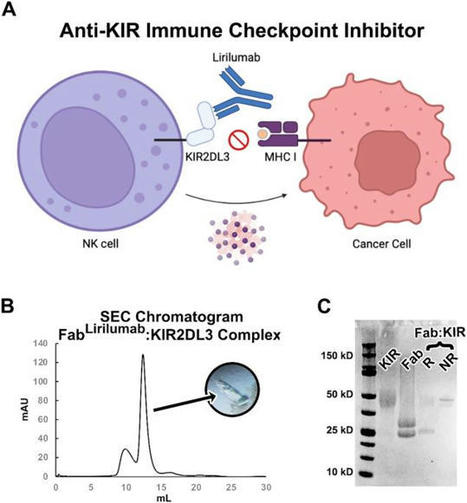
#Immunotherapy | How #Lirilumab #KIR-tails an #ImmuneCheckpoint | New OPEN ACCESS Study by Nicholas Lorig-Roach et al Unveils General Concepts for the…
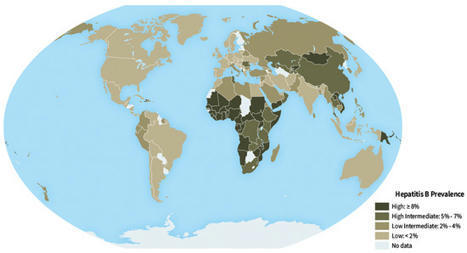
Hepatitis B virus (HBV) infection is a global public health problem that is closely related to liver cirrhosis and hepatocellular carcinoma (HCC). The prevalence of acute and chronic HBV infection, liver cirrhosis, and HCC has significantly decreased as a result of the introduction of universal...
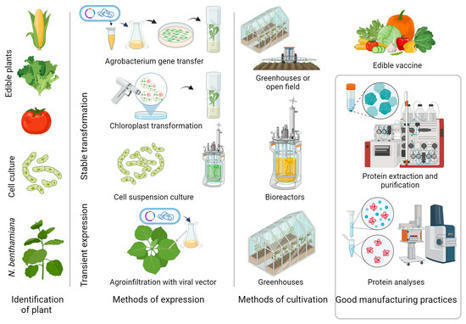
Plants are increasingly used for the production of high-quality biological molecules for use as pharmaceuticals and biomaterials in industry. Plants have proved that they can produce life-saving therapeutic proteins (Elelyso™—Gaucher’s disease treatment, ZMapp™—anti-Ebola monoclonal antibodies,...
Engineered immune cells have given 15 people with once-debilitating autoimmune disorders a new lease on life, free from fresh symptoms or treatments. The results raise hopes that the approach — called CAR-T-cell therapy — might one day be extended to a variety of other conditions fuelled by rogue immune cells that produce antibodies against the body’s own tissues. All 15 participants, who each had one of three autoimmune conditions, have remained disease-free or nearly so since their treatment, according to data presented on 9 December at the American Society of Hematology meeting in San Diego, California. The first participants were treated more than two years ago. These successes, although preliminary, have been electric, says Marco Ruella, an oncologist at the University of Pennsylvania in Philadelphia. “We’re all excited,” he says. “There’s a lot of potential.” Bespoke immune cells CAR-T therapies harness the immune players called T cells. T cells are removed from the person being treated, genetically engineered to produce proteins called chimeric antigen receptors (CARs) and then reintroduced to the person’s body. In many therapies, the T cells are tailored to recognize a protein made by immune cells called B cells. When reintroduced, the CAR T cells will target the B cells for destruction — a useful feature for treating cancers caused by abnormal B cells. B cells also drive some autoimmune disorders by making antibodies that attack healthy tissue. In 2019, researchers showed that CAR T cells that recognize B cells reduced symptoms in mice with a disease similar to lupus, an autoimmune disorder that affects a variety of organs1. Around the same time, researchers at University Hospital Erlangen in Germany were setting up their own CAR-T centre to provide cancer treatment. During a meeting at the centre, a rheumatologist asked the cancer specialists for advice about a young woman with a form of lupus called systemic lupus erythematosus. Several of her organs were failing; her doctors estimated that she did not have long to live. The young woman insisted that they try something new. High-risk approach The team thought of the mouse study but baulked at trying it in people. CAR-T therapy can have severe side effects, and recipients must first undergo intensive chemotherapy that kills off many of their existing immune cells. “At the beginning we were quite scared,” said team member Fabian Müller, an oncologist at the Friedrich–Alexander University of Erlangen–Nuremberg, at a press conference before he presented the work at the San Diego meeting. The woman was adamant that they try. That first participant — and the others who followed — experienced relatively minor adverse effects, Müller reported at the conference. The Erlangen team eventually used the method to treat two other autoimmune disorders: systemic sclerosis and idiopathic inflammatory myositis. The successes continued. Other groups have since taken up the approach and reported similar results. Earlier this month, another team added a fourth autoimmune disorder called myasthenia gravis to the list of successes2. Researchers are beginning to wonder how long the final list will be. “We’re just at the beginning,” says Marcela Maus, who designs CAR-T therapies against cancer at Massachusetts General Hospital in Boston. “There is so much that can be done that was unthinkable just a decade ago.” At this stage, however, it’s unclear how much of this success is due to the CAR-T therapy as opposed to the chemotherapy that killed many of the participants’ pre-existing immune cells, cautions Ruella. That might have helped to wipe out the errant B cells. For now, Müller lapses into a dreamy smile as he marvels over the remarkable recoveries he has seen: the man who struggled to walk 10 metres before his treatment and now routinely walks 10 kilometres around town, for example. “These are young people that have been spending more time with their doctors than with their friends,” he says. “They would describe their breakfast as a handful of pills that they are just shoving in.” “And it’s all gone,” he says. “From the physician perspective, it’s pretty much the most pleasing thing.” December 12, 2023 https://doi.org/10.1038/d41586-023-03968-6
Via Juan Lama
PD-1 (Programmed Cell Death Protein-1) and PD-L1 (Programmed Cell Death Ligand-1) play a crucial role in regulating the immune system and preventing autoimmunity. Cancer cells can manipulate this system, allowing them to escape immune detection and promote tumor growth. Therapies targeting the PD-1/PD-L1 pathway have transformed cancer treatment and have demonstrated significant effectiveness against various cancer types. This study delves into the structure and signaling dynamics of PD-1 and its ligands PD-L1/PD-L2, the diverse PD-1/PD-L1 inhibitors and their efficacy, and the resistance observed in some patients. Furthermore, this study explored the challenges associated with the PD-1/PD-L1 inhibitor treatment approach. Recent advancements in the combination of immunotherapy with chemotherapy, radiation, and surgical procedures to enhance patient outcomes have also been highlighted. Overall, this study offers an in-depth overview of the significance of PD-1/PD-L1 in cancer immunotherapy and its future implications in oncology.
CAR T cell therapy, a powerful type of immunotherapy, has begun to revolutionize cancer treatment. Pioneered at Memorial Sloan Kettering Cancer Center (MSK), the therapy involves engineering a patient's T cells so they recognize and attack cancer cells.
|



 Your new post is loading...
Your new post is loading...