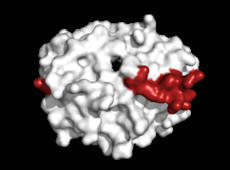
The new observation, made by UNC School of Medicine’s Stephan Moll, MD, and Jacquelyn Baskin-Miller, MD, suggests that a life-threatening blood clotting disorder can be caused by an infection with adenovirus, one of the most common respiratory viruses in pediatric and adult patients.
CHAPEL HILL, N.C. – Platelets, or thrombocytes, are specialized cellular fragments that form blood clots when we get scrapes and traumatic injuries. Viral infections, autoimmune disease, and other conditions can cause platelet levels to drop throughout the body, termed thrombocytopenia. After a robust clinical and research collaboration, Stephan Moll, MD, and Jacquelyn Baskin-Miller, MD, both in the UNC School of Medicine, have linked adenovirus infection with a rare blood clotting disorder. This is the first time that the common respiratory virus, which causes mild cold-and flu-like symptoms, has been reported to be associated with blood clots and severe thrombocytopenia. “This adenovirus-associated disorder is now one of four recognized anti-PF4 disorders,” said Moll, professor of medicine in the Department of Medicine’s Division of Hematology. “We hope that our findings will lead to earlier diagnosis, appropriate and optimized treatment, and better outcomes in patients who develop this life-threatening disorder.” Their new observation, which was published in the New England Journal of Medicine, sheds new light on the virus and its role in causing an anti-platelet factor 4 disorder. Additionally, the discovery opens a whole new door for research, as many questions remain as to how and why this condition occurs – and who is most likely to develop the disorder.
HIT, VITT, and “Spontaneous HIT”
Antibodies are large Y-shaped proteins that can stick to the surface of bacteria and other “foreign” substances, flagging them for destruction by the immune system or neutralizing the threat directly. In anti-PF4 disorders, the person’s immune system makes antibodies against platelet factor-4 (PF4), a protein that is released by platelets. When an antibody forms against PF4 and binds to it, this can trigger the activation and rapid removal of platelets in the bloodstream, leading to blood clotting and low platelets, respectively. Sometimes, the formation of anti-PF4 antibodies is triggered by a patient’s exposure to heparin, called heparin-induced thrombocytopenia (HIT), and sometimes it occurs as an autoimmune condition without heparin exposure, which is referred to as “spontaneous HIT.” In the last three years, thrombocytopenia has been shown to rarely occur after injection with COVID-19 vaccines that are made with inactivated pieces of an adeno–viral vector. These vaccines are different than the ones made in the United States, such as those by Moderna and Pfizer. The condition is referred to as vaccine-induced immune thrombotic thrombocytopenia (VITT).
The Road to Discovery
The road to the discovery started when a young child, who had been diagnosed as an outpatient with adenovirus infection, had to be admitted to the hospital with an aggressive blood clot forming in his brain (called cerebral sinus vein thrombosis) and severe thrombocytopenia. Doctors determined that they hadn’t been exposed to heparin or the adeno-vector COVID-19 vaccination, the classical triggers for HIT and VITT. “The intensive care unit physicians, the neuro-intensivist, and hematology group were working around the clock to determine next steps in the care for this young child,” said Baskin-Miller. “They weren’t responding to therapy and were progressing quickly. We had questioned whether it could have been linked to the adenovirus considering the vaccine data, but there was nothing in the literature at that time to suggest it.” The collaborative clinical effort to help the patient expanded: Baskin-Miller reached out to Moll, who is an expert in thrombosis and has various connections throughout the field. To Moll, it looked like the pediatric patient could have “spontaneous HIT”. They then tested for the HIT platelet activating antibody, which came back positive.
Collaboration is Key
Moll reached out to, Theodore E. Warkentin, MD, a professor of pathology and molecular medicine at McMaster University in Hamilton, Ontario, who has been researching anti-PF4 disorders for three decades, to hear if he was aware of an association between adenoviral infection and spontaneous HIT. Warkentin, who is one of the premier international anti-PF-4 disorders researcher, wasn’t aware of the condition. Around the same time, Moll received a phone call from Alison L. Raybould, MD, a hematologist-oncologist in Richmond, Virginia, a previous trainee from UNC. She was seeing a patient who had multiple blood clots, a stroke and heart attack, arm and leg deep-vein thromboses (DVT), and severe thrombocytopenia. The patient had not been exposed to heparin or vaccines. However, this patient’s severe illness had also started with viral symptoms of cough and fever, and she had tested positive for adenoviral infection. Testing for an anti-PF4 antibody also turned out to be positive. To help clarify the diagnoses of the two patients, Warkentin immediately offered to further test the patients’ blood and samples were directly to his laboratory in the Hamilton General Hospital for further study. They confirmed that the antibodies were targeting platelet factor 4, much like the HIT antibodies. Surprisingly, the antibody resembled that of the VITT and bound to PF4 in the same region as VITT antibodies do. They concluded that both the patients had “spontaneous HIT” or a VITT-like disorder, associated with an adenovirus infection.
More Questions
Following such a groundbreaking conclusion, Moll and colleagues are now left with many questions about the prevalence of the new anti-PF4 disorder, whether the condition can be caused by other viruses, and why this condition doesn’t occur with every infection with adenovirus. They also wonder what preventative or treatment measures can be made to help patients who develop the new, potentially deadly anti-PF4 disorder. “How common is the disorder?” asked Moll. “What degree of thrombocytopenia raises the threshold to test for anti-PF4 antibodies? And then finally, how do we best treat these patients to optimize the chance that they will survive such a potentially deadly disease?”
Media contact: Kendall Daniels, Communications Specialist, UNC Health | UNC School of Medicine
Original research published in New England J. Medic. (August 10, 2023):


 Your new post is loading...
Your new post is loading...