Your new post is loading...

|
Scooped by
Juan Lama
|
Scientists have studied the immune system of a man in Germany who claims to have received 217 Covid-19 vaccines. The researchers were concerned that the man's immune system might have become exhausted with such frequent and numerous challenges with vaccines against the same virus, but they actually found the opposite. Their analysis published in the journal Lancet Infectious Diseases found that not only did the man, 62, have a fully functional immune system, there were indicators that his immunity against the virus that causes Covid-19 was better than people who had just received a normal number of vaccines. There is official evidence of the man having 134 Covid-19 vaccines of eight different types including mRNA vaccines over a nine-month period, although the man claims the amount to be 217 in total over 29 months...

|
Scooped by
Juan Lama
|
The SARS-CoV-2 Omicron variant and its numerous sub-lineages have exhibited a striking ability to evade humoral immune responses induced by prior vaccination or infection. The Food and Drug Administration (FDA) has recently granted Emergency Use Authorizations (EUAs) to new bivalent formulations of the original Moderna and Pfizer mRNA SARS-CoV-2 vaccines that target both the ancestral strain as well as the Omicron BA.4/BA.5 variant. Despite their widespread use as a vaccine boost, little is known about the antibody responses induced in humans. Here, we collected sera from several clinical cohorts: individuals after three or four doses of the original monovalent mRNA vaccines, individuals receiving the new bivalent vaccines as a fourth dose, and individuals with BA.4/BA.5 breakthrough infection following mRNA vaccination. Using pseudovirus neutralization assays, these sera were tested for neutralization against an ancestral SARS-CoV-2 strain, several Omicron sub-lineages, and several related sarbecoviruses. At ~3-5 weeks post booster shot, individuals who received a fourth vaccine dose with a bivalent mRNA vaccine targeting BA.4/BA.5 had similar neutralizing antibody titers as those receiving a fourth monovalent mRNA vaccine against all SARS-CoV-2 variants tested, including BA.4/BA.5. Those who received a fourth monovalent vaccine dose had a slightly higher neutralizing antibody titers than those who received the bivalent vaccine against three related sarbecoviruses: SARS-CoV, GD-Pangolin, and WIV1. When given as a fourth dose, a bivalent mRNA vaccine targeting Omicron BA.4/BA.5 and an ancestral SARS-CoV-2 strain did not induce superior neutralizing antibody responses in humans, at the time period tested, compared to the original monovalent vaccine formulation. Preprint available in bioRxiv (Oct. 24, 2022): https://www.biorxiv.org/content/10.1101/2022.10.22.513349v1 https://doi.org/10.1101/2022.10.22.513349

|
Scooped by
Juan Lama
|
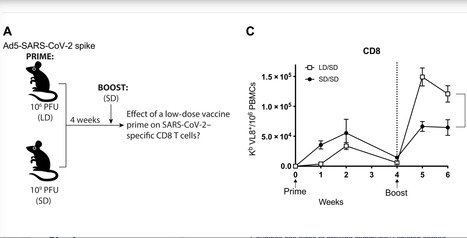
SARS-CoV-2 has caused a global pandemic that has infected more than 250 million people worldwide. Although several vaccine candidates have received emergency use authorization, there is still limited knowledge on how vaccine dosing affects immune responses. We performed mechanistic studies in mice to understand how the priming dose of an adenovirus-based SARS-CoV-2 vaccine affects long-term immunity to SARS-CoV-2. We first primed C57BL/6 mice with an adenovirus serotype 5 vaccine encoding the SARS-CoV-2 spike protein, similar to that used in the CanSino and Sputnik V vaccines. The vaccine prime was administered at either a standard dose or 1000-fold lower dose, followed by a boost with the standard dose 4 weeks later. Initially, the low dose prime induced lower immune responses relative to the standard dose prime. However, the low dose prime elicited immune responses that were qualitatively superior and, upon boosting, exhibited substantially more potent recall and functional capacity. We also report similar effects with a simian immunodeficiency virus (SIV) vaccine. These findings show an unexpected advantage of fractionating vaccine prime doses, warranting a reevaluation of vaccine trial protocols for SARS-CoV-2 and other pathogens. Published in Science Immunology: https://doi.org/10.1126/sciimmunol.abi8635
|

|
Scooped by
Juan Lama
|
In a recent article published in the journal PNAS, researchers in China provide evidence that a novel vaccine candidate known as CF501/RBD-Fc robustly neutralized severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Omicron subvariants BQ.1.1 and XBB in a rhesus macaque animal model. This vaccine comprised the human immunoglobulin G (hIgG) fraction, crystallizable (Fc)-conjugated receptor-binding domain (RBD) of the SARS-CoV-2 ancestral WA1 strain, in combination with a novel stimulator of interferon genes (STING) agonist-based adjuvant called CF501. The study findings confirm that CF501/RBD-Fc induced highly potent and persistent broad-neutralizing antibody (bnAb) responses against several SARS-CoV-2 variants, including Omicron subvariants. Background Due to their exceptional immune-evasion properties, Omicron subvariants pose a significant challenge to current coronavirus disease 2019 (COVID-19) vaccines. For example, the BA.5 subvariant is resistant to neutralization, even after four doses of a messenger ribonucleic acid (mRNA) vaccine. Moreover, the newly emerged XBB Omicron subvariant remains unneutralized by nAbs induced by a booster dose of the bivalent vaccine containing the mRNA sequences of the Omicron BA.5 and ancestral spike (S) proteins. Previous studies using the pseudovirus neutralization assay have shown that, as compared to the ancestral strain D614G, XBB is up to 155-fold more resistant to nAbs in vaccinee sera. Thus, there remains an urgent need for a pan-sarbecovirus vaccine with the ability to neutralize current and yet-to-emerge SARS-CoV-2 variants. About the study In the present study, researchers administered three doses of CF501/RBD-Fc or Alum/RBD-Fc-based vaccines in two groups, each comprised of three rhesus macaques. Sera was subsequently collected to evaluate RBD-binding IgG and nAb titers through the use of an enzyme-linked immunosorbent assay (ELISA) and virus neutralization assays. The researchers also tested whether sera from immunized rhesus macaques could neutralize pseudotyped Omicron subvariants. Each test animal's parameters were correlated by pairwise comparisons to assess the association between nAb and binding antibodies specific to Omicron subvariants XBB and BQ.1.1. Results At day 28, after two vaccine doses, endpoint RBD-specific IgG titers against Omicron subvariants in the CF501/RBD-Fc group ranged between 512,000 and 1,792,000. These values were nearly three- to 28-fold higher than that of the Alum/RBD-Fc group. Although the endpoint titers gradually declined, they remained relatively stable and higher in the CF501/RBD-Fc group as compared to the Alum/RBD-Fc group. The magnitude of the RBD-binding antibodies remained consistently higher in the CF501/RBD-Fc group after three vaccine doses and remained high until day 191 following the first vaccination. The 50% neutralizing titers (NT50) of bnAbs in sera from CF501/RBD-Fc macaques were much higher than those in the Alum/RBD-Fc group against all pseudotyped viruses, with NT50 values of 436 and 313 against BQ.1.1 and XBB at day 28, respectively. Cross-neutralizing bnAb titers in the CF501/RBD-Fc group continued to increase after the third vaccination, with day 122 NT50 values of 2,118 and 2,526 against BQ.1.1 and XBB after receipt of the first vaccine dose, respectively. These titers also increased in the Alum/RBD-Fc group after three vaccine doses; however, these values marginally surged against BQ.1.1 and XBB. Eventually, NT50 values declined in both groups. The third vaccine dose did not elicit increased NT50 titers against D614G but drastically increased bnAb titers against the Omicron subvariants. Although their NT50 against BQ.1.1 and XBB declined by 26.9- and 22.5-fold relative to D614G, respectively, these bnAbs effectively neutralized BQ.1.1 and XBB infection. Virus neutralization assay results also showed that CF501/RBD-Fc sera potently neutralized authentic BA.2.2 infection as compared to Alum/RBD-Fc sera. Immunofluorescence assay also confirmed that sera from CF501/RBD-Fc group potently inhibited Omicron BA.2.2 replication. Conclusions Overall, the study findings indicate that the CF501 adjuvant stimulated the conservative but nondominant RBD epitopes for generating bnAbs against pan-sarbecovirus vaccines. Thus, the researchers recommend replacing the adjuvant in the first-generation COVID-19 subunit vaccines with CF501 for next-generation booster vaccinations. This strategy might enhance the immune responses against SARS-CoV-2 Omicron subvariants BQ.1.1 and XBB, as well as future SARS-CoV-2 variants that have yet to emerge. Research cited published (March 10, 2023) in PNAS: https://doi.org/10.1073/pnas.2221713120

|
Scooped by
Juan Lama
|
The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, has launched an early-stage clinical trial evaluating an investigational vaccine to prevent infection with Nipah virus. The experimental vaccine is manufactured by Moderna, Inc., Cambridge, Massachusetts, and was developed in collaboration with NIAID’s Vaccine Research Center. It is based on a messenger RNA (mRNA) platform—a technology used in several approved COVID-19 vaccines. NIAID is sponsoring the Phase 1 clinical study, which is being conducted at the NIH Clinical Center in Bethesda, Maryland. Nipah virus infection is a zoonotic disease, meaning that it is spread between animals and people. Fruit bats are the natural host for the virus. The first known Nipah outbreak occurred in 1998 in Malaysia and Singapore and resulted in 265 human cases and 105 deaths, and caused significant economic damage to the swine industry there. Since 1999, outbreaks have occurred annually in Asia, primarily in Bangladesh and India. The virus can cause mild-to-severe disease rapidly progressing from respiratory infection symptoms to encephalitis (brain swelling) leading to coma or death. An estimated 40% to 75% of people infected with Nipah virus die. Although most cases are transmitted via animals, person-to-person transmission can occur. Currently, there is no licensed vaccine or treatment for Nipah virus infection. “Nipah virus poses a considerable pandemic threat because it mutates relatively easily, causes disease in a wide range of mammals, can transmit from person-to-person, and kills a large percentage of the people it infects,” said NIAID Director Anthony S. Fauci, M.D. “The need for a preventive Nipah virus vaccine is significant.” NIAID’s Pandemic Preparedness Plan, published earlier this year, established a framework to study viruses of pandemic potential and prioritize research on prototype pathogens, such as Nipah virus. This is the first clinical trial using the prototype pathogen approach since the plan’s publication. The experimental mRNA-1215 Nipah virus vaccine will be tested in a dose-escalation clinical trial to evaluate its safety, tolerability, and ability to generate an immune response in 40 healthy adults ages 18 to 60 years. Specifically, four groups of 10 participants each will receive two doses of the investigational vaccine via injection in the shoulder muscle four or 12 weeks apart. Group one (10 participants) will receive two 25-microgram (mcg) injections; group two will receive two 50-mcg injections; and group three will receive two 100-mcg injections, each four weeks apart. The vaccine dose for the fourth group of participants will be determined based on an interim analysis of the results from the three previous groups. The fourth group will receive two injections 12 weeks apart. Study participants will be evaluated through clinical observation and blood collection at specified times throughout the study and will be followed by clinical study staff through 52 weeks following their final vaccination. For more information about the clinical trial, visit ClinicalTrials.gov using the study identifier NCT05398796. NIAID conducts and supports research—at NIH, throughout the United States, and worldwide—to study the causes of infectious and immune-mediated diseases, and to develop better means of preventing, diagnosing, and treating these illnesses. News releases, fact sheets and other NIAID-related materials are available on the NIAID website.

|
Scooped by
Juan Lama
|
Malaria kills about 500,000 people each year, about half of them children in Africa. The new vaccine isn’t perfect, but it will help turn the tide, experts said. The world has gained a new weapon in the war on malaria, among the oldest known and deadliest of infectious diseases: the first vaccine shown to help prevent the disease. By one estimate, it will save tens of thousands of children each year. Malaria kills about half a million people each year, nearly all of them in sub-Saharan Africa — including 260,000 children under 5. The new vaccine, made by GlaxoSmithKline, rouses a child’s immune system to thwart Plasmodium falciparum, the deadliest of five malaria pathogens and the most prevalent in Africa. The World Health Organization on Wednesday endorsed the vaccine, the first step in a process that should lead to wide distribution in poor countries. To have a malaria vaccine that is safe, moderately effective and ready for distribution is “a historic event,” said Dr. Pedro Alonso, director of the W.H.O.’s global malaria program. Malaria is rare in the developed world. There are just 2,000 cases in the United States each year, mostly among travelers returning from countries in which the disease is endemic. The vaccine, called Mosquirix, is not just a first for malaria — it is the first developed for any parasitic disease. Parasites are much more complex than viruses or bacteria, and the quest for a malaria vaccine has been underway for a hundred years. “It’s a huge jump from the science perspective to have a first-generation vaccine against a human parasite,” Dr. Alonso said. In clinical trials, the vaccine had an efficacy of about 50 percent against severe malaria in the first year, but the figure dropped close to zero by the fourth year. And the trials did not directly measure the vaccine’s impact on deaths, which has led some experts to question whether it is a worthwhileinvestment in countries with countless other intractable problems. But severe malaria accounts for up to half of malaria deaths and is considered “a reliable proximal indicator of mortality,” said Dr. Mary Hamel, who leads the W.H.O.’s malaria vaccine implementation program. “I do expect we will see that impact.” A modeling study last year estimated that if the vaccine were rolled out to countries with the highest incidence of malaria, it could prevent 5.4 million cases and 23,000 deaths in children younger than 5 each year. A recent trial of the vaccine in combination with preventive drugs given to children during high-transmission seasons found that the dual approach was much more effective at preventing severe disease, hospitalization and death than either method alone. The malaria parasite, carried by mosquitoes, is a particularly insidious enemy, because it can strike the same person over and over. In many parts of sub-Saharan Africa, even those where most people sleep under insecticide-treated bed nets, children have on average six malaria episodes a year. Even when the disease is not fatal, the repeated assault on their bodies can permanently alter the immune system, leaving them weak and vulnerable to other pathogens. Malaria research is littered with vaccine candidates that never made it past clinical trials. Bed nets, the most widespread preventive measure, cut malaria deaths in children under 5 only by about 20 percent. Against that backdrop, the new vaccine, even with modest efficacy, is the best new development in the fight against the disease in decades, some experts said. “Progress against malaria has really stalled over the last five or six years, particularly in some of the hardest hit countries in the world,” said Ashley Birkett, who heads malaria programs at PATH, a nonprofit organization focused on global health. With the new vaccine, “there’s potential for very, very significant impact there,” Dr. Birkett said. Mosquirix is given in three doses between ages 5 and 17 months, and a fourth dose roughly 18 months later. Following clinical trials, the vaccine was tried out in three countries — Kenya, Malawi and Ghana — where it was incorporated into routine immunization programs. More than 2.3 million doses have been administered in those countries, reaching more than 800,000 children. That bumped up the percentage of children protected against malaria in some way to more than 90 percent, from less than 70 percent, Dr. Hamel said. “The ability to reduce inequities in access to malaria prevention — that’s important,” Dr. Hamel said. “It was impressive to see that this could reach children who are currently not being protected.” It took years to create an efficient system to distribute insecticide-treated bed nets to families. By contrast, including Mosquirix among routine immunizations made it surprisingly easy to distribute, Dr. Hamel added — even in the midst of the coronavirus pandemic, which prompted lockdowns and disrupted supply chains. “We aren’t going to have to spend a decade trying to figure out how to get this to children,” she said. This week, a working group of independent experts in malaria, child health epidemiology and statistics, as well as the W.H.O.’s vaccine advisory group, met to review data from the pilot programs and to make their formal recommendation to Dr. Tedros Adhanom Ghebreyesus, director-general of the W.H.O. “We still have a very long road to travel, but this is a long stride down that road,” Dr. Tedros said at a news conference on Wednesday. The next step is for Gavi, the global vaccine alliance, to determine that the vaccine is a worthwhile investment. If the organization’s board approves the vaccine — not guaranteed, given the vaccine’s moderate efficacy and the many competing priorities — Gavi will purchase the vaccine for countries that request it, a process that is expected to take at least a year. But as with Covid-19, problems with vaccine production and supply could considerably delay progress. And the pandemic has also diverted resources and attention from other diseases, said Deepali Patel, who leads malaria vaccine programs at Gavi. “Covid is a big unknown in the room in terms of where capacity is currently in countries, and rolling out Covid-19 vaccines is a huge effort,” Ms. Patel said. “We’re really going to have to see how the pandemic unfolds next year in terms of when countries will be ready to pick up all of these other priorities.”
|


 Your new post is loading...
Your new post is loading...