 Your new post is loading...
 Your new post is loading...
The bioengineered immune players called CAR T cells last longer and work better if pumped up with a large dose of a protein that makes them resemble stem cells.
Magnetic resonance imaging (MRI) and lumbar puncture (LP) may not always be necessary for diagnosing and managing a serious neurological complication associated with CAR T-cell therapy, according to a new Blood Advances study.
study evaluates senolytic CAR T-cell therapy targeting uPAR-positive cells in aged mice, showing its effectiveness in mitigating age-related metabolic dysfunction and offering a potential long-lasting treatment for aging-associated conditions.
Immunotherapy using modified chimeric antigen receptor (CAR) T cells has greatly improved survival rates for pediatric patients with relapsed and recurrent leukemia.
Vα24-invariant natural killer T cells (NKTs) have anti-tumor properties that can be enhanced by chimeric antigen receptors (CARs). Here we report updated interim results from the first-in-human phase 1 evaluation of autologous NKTs co-expressing a GD2-specific CAR with interleukin 15 (IL15) (GD2-CAR.15) in 12 children with neuroblastoma (NB). The primary objectives were safety and determination of maximum tolerated dose (MTD). The anti-tumor activity of GD2-CAR.15 NKTs was assessed as a secondary objective. Immune response evaluation was an additional objective. No dose-limiting toxicities occurred; one patient experienced grade 2 cytokine release syndrome that was resolved by tocilizumab. The MTD was not reached. The objective response rate was 25% (3/12), including two partial responses and one complete response. The frequency of CD62L+NKTs in products correlated with CAR-NKT expansion in patients and was higher in responders (n = 5; objective response or stable disease with reduction in tumor burden) than non-responders (n = 7). BTG1 (BTG anti-proliferation factor 1) expression was upregulated in peripheral GD2-CAR.15 NKTs and is a key driver of hyporesponsiveness in exhausted NKT and T cells. GD2-CAR.15 NKTs with BTG1 knockdown eliminated metastatic NB in a mouse model. We conclude that GD2-CAR.15 NKTs are safe and can mediate objective responses in patients with NB. Additionally, their anti-tumor activity may be enhanced by targeting BTG1. ClinicalTrials.gov registration: NCT03294954 . In updated results from a phase 1 trial of GD2-specific CAR-NKT cells in patients with neuroblastoma, no dose-limiting toxicities were observed across multiple dose levels; the maximum tolerated dose was not reached; and there was evidence of anti-tumor activity.
Chimeric antigen receptor T-cell (CAR-T) therapy has transformed cancer treatment, yet relatively few studies have investigated the impact of the therapy on longitudinal patient quality of life – an aspect of care that often suffers from receiving traditional intensive cancer medications, such as chemotherapy.
With a slew of tools to trick out immune cells, researchers are expanding the repertoire of CAR-T therapies.
In two separate studies, researchers demonstrate how synthetic biology can be used to tackle a difficult issue in cancer immunotherapy: the way immunotherapy-related approaches focused on short-term killing of tumor cells may fail to eradicate tumors because growth of tumors happens on longer timescales.
A new approach to cancer immunotherapy that uses one type of immune cell to kill another-;rather than directly attacking the cancer-;provokes a robust anti-tumor immune response that shrinks ovarian, lung, and pancreatic tumors in preclinical disease models, according to researchers at the Icahn School of Medicine at Mount Sinai in New York.
The use of broad-spectrum antibiotics in the month leading up to chimeric antigen receptor T-cell therapy led to poorer outcomes and increased treatment-related toxicities, study results showed.Certain types of gut bacteria had an impact on treatment efficacy and related toxicities among patients who received CAR-T for non-Hodgkin lymphoma or acute lymphoblastic leukemia, researchers reported.
Synthetic receptor signalling has the potential to endow adoptively transferred T cells with new functions that overcome major barriers in the treatment of solid tumours, including the need for conditioning chemotherapy1,2. Here we designed chimeric receptors that have an orthogonal IL-2 receptor extracellular domain (ECD) fused with the intracellular domain (ICD) of receptors for common γ-chain (γc) cytokines IL-4, IL-7, IL-9 and IL-21 such that the orthogonal IL-2 cytokine elicits the corresponding γc cytokine signal. Of these, T cells that signal through the chimeric orthogonal IL-2Rβ-ECD–IL-9R-ICD (o9R) are distinguished by the concomitant activation of STAT1, STAT3 and STAT5 and assume characteristics of stem cell memory and effector T cells. Compared to o2R T cells, o9R T cells have superior anti-tumour efficacy in two recalcitrant syngeneic mouse solid tumour models of melanoma and pancreatic cancer and are effective even in the absence of conditioning lymphodepletion. Therefore, by repurposing IL-9R signalling using a chimeric orthogonal cytokine receptor, T cells gain new functions, and this results in improved anti-tumour activity for hard-to-treat solid tumours. Synthetic chimeric orthogonal IL-2 receptors that incorporate the intracellular domain of receptors for other γ-chain cytokines such as IL-9 can reroute orthogonal signalling and alter the phenotype of T cells to improve anti-tumour responses.
Despite their clinical success, chimeric antigen receptor (CAR)-T cell therapies for B cell malignancies are limited by lengthy, costly and labor-intensive ex vivo manufacturing procedures that might lead to cell products with heterogeneous composition. Here we describe an implantable Multifunctional Alginate Scaffold for T Cell Engineering and Release (MASTER) that streamlines in vivo CAR-T cell manufacturing and reduces processing time to a single day. When seeded with human peripheral blood mononuclear cells and CD19-encoding retroviral particles, MASTER provides the appropriate interface for viral vector-mediated gene transfer and, after subcutaneous implantation, mediates the release of functional CAR-T cells in mice. We further demonstrate that in vivo-generated CAR-T cells enter the bloodstream and control distal tumor growth in a mouse xenograft model of lymphoma, showing greater persistence than conventional CAR-T cells. MASTER promises to transform CAR-T cell therapy by fast-tracking manufacture and potentially reducing the complexity and resources needed for provision of this type of therapy. Implantable scaffolds rapidly generate and release anti-tumor CAR-T cells in mice.
A chimeric antigen receptor T-cell therapy induced high response rates across a range of B-cell malignancies, according to phase 1 study results presented at ASH Annual Meeting and Exposition.An interim analysis showed a 93% overall response rate among 15 patients with relapsed or refractory follicular lymphoma who received the investigational therapy.
|
Treatment with a next-generation CAR T-cell agent displayed early efficacy in a small group of patients with glioblastoma.
Targeting two brain tumor-associated proteins-;rather than one-;with CAR T cell therapy shows promise as a strategy for reducing solid tumor growth in patients with recurrent glioblastoma (GBM), an aggressive form of brain cancer, according to early results from the first six patients treated in an ongoing Phase I clinical trial led by researchers from the Perelman School of Medicine at the University of Pennsylvania and Penn Medicine's Abramson Cancer Center.
At EPFL's School of Engineering, Professor Li Tang's Laboratory of Biomaterials for Immunoengineering has made significant strides in cancer treatment research.
A broad new strategy could hold hope for treating virtually all blood cancers with CAR T cell therapy, which is currently approved for five subtypes of blood cancer.
Adoptive transfer of genetically engineered chimeric antigen receptor (CAR) T cells is becoming a promising treatment option for hematological malignancies. However, T cell immunotherapies have mostly failed in individuals with solid tumors. Here, with a CRISPR–Cas9 pooled library, we performed an in vivo targeted loss-of-function screen and identified ST3 β-galactoside α-2,3-sialyltransferase 1 (ST3GAL1) as a negative regulator of the cancer-specific migration of CAR T cells. Analysis of glycosylated proteins revealed that CD18 is a major effector of ST3GAL1 in activated CD8+ T cells. ST3GAL1-mediated glycosylation induces the spontaneous nonspecific tissue sequestration of T cells by altering lymphocyte function-associated antigen-1 (LFA-1) endocytic recycling. Engineered CAR T cells with enhanced expression of βII-spectrin, a central LFA-1-associated cytoskeleton molecule, reversed ST3GAL1-mediated nonspecific T cell migration and reduced tumor growth in mice by improving tumor-specific homing of CAR T cells. These findings identify the ST3GAL1–βII-spectrin axis as a major cell-intrinsic program for cancer-targeting CAR T cell migration and as a promising strategy for effective T cell immunotherapy. CAR T cell success requires targeting tumors, but these cells can get trapped in other tissues, such as in the lungs, where they can cause pathology. Here, the authors use a loss-of-function CRISPR screen to identify regulators of CAR T cell tumor trafficking and engineer CAR T cells accordingly to overcome this limitation.
Pancreatic cancer is an incurable form of cancer, and gene therapies are currently in clinical testing to treat this deadly disease. A comprehensive review of the gene and cell biotherapies in development to combat pancreatic cancer is published in the peer-reviewed journal Human Gene Therapy.
Researchers at UCSF have developed a novel, potentially life-saving approach that may prevent antibodies from triggering immune rejection of engineered therapeutic and transplant cells.
Precision-controlled CAR-T-cell immunotherapies could be used to tackle a range of tumour types.
‘Most complicated therapy ever’ tailors bespoke, genome-edited immune cells to attack tumours.
Scientists at St. Jude Children's Research Hospital identified a molecular mechanism that in a preclinical study unlocked the promise of CAR T–cell therapy for treatment of solid tumors.
Genetically engineered immune cells successfully target the specific cancer cells that may be responsible for relapse of acute myeloid leukemia (AML), a type of blood cancer, and proved effective in animal models of the disease, according to a preclinical study by investigators at Weill Cornell Medicine.
Clinicians who treat patients with chimeric antigen receptor T cells have become adept at identifying and treating acute neurotoxicity, a common adverse event associated with the therapy.Researchers from Mount Sinai published a case study in Nature Medicine about a patient who developed neurocognitive and hypokinetic movement disorder with features of Parkinson’s disease after receiving
|



 Your new post is loading...
Your new post is loading...

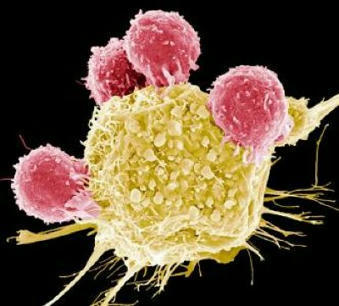



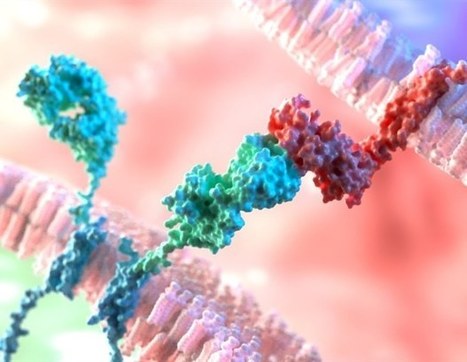

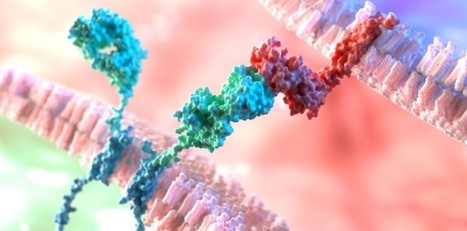










Keeping cells active long enough to eliminate cancer has proved difficult, particularly in solid tumors such as those of the breast and lung. Scientists are therefore looking for better ways to help CAR T cells multiply faster and last longer in the body. To this end, researchers compared samples of CAR T cells used to treat people with leukemia. They analyzed the role of cellular proteins that regulate gene activity and serve as master switches in T cells. They discovered a set of 41 genes that were more active in CAR T cells associated with a good response to treatment than in cells associated with a poor response. All 41 genes appeared to be regulated by a master protein called FOXO1. The researchers then engineered the CAR T cells to produce more FOXO1 than usual. Gene activity in these cells began to resemble that of memory T stem cells, which recognize cancer and respond quickly to it. The researchers then injected the modified cells into mice with different types of cancer. Extra FOXO1 enabled the CAR T cells to better reduce both solid tumors and blood cancers. Moreover, another team also reached the same conclusion by working on gene activity analysis in CAR T cells and also discovered that IL-15 activated genes associated with FOXO1.