The transcription factors nuclear factor of activated T cells (NFAT) and activator protein 1 (AP-1; Fos–Jun) cooperate to promote the effector functions of T cells, but NFAT in the absence of AP-1 imposes a negative feedback program of T cell hyporesponsiveness (exhaustion). Here, we show that basic leucine zipper ATF-like transcription factor (BATF) and interferon regulatory factor 4 (IRF4) cooperate to counter T cell exhaustion in mouse tumor models. Overexpression of BATF in CD8+ T cells expressing a chimeric antigen receptor (CAR) promoted the survival and expansion of tumor-infiltrating CAR T cells, increased the production of effector cytokines, decreased the expression of inhibitory receptors and the exhaustion-associated transcription factor TOX and supported the generation of long-lived memory T cells that controlled tumor recurrence. These responses were dependent on BATF–IRF interaction, since cells expressing a BATF variant unable to interact with IRF4 did not survive in tumors and did not effectively delay tumor growth. BATF may improve the antitumor responses of CAR T cells by skewing their phenotypes and transcriptional profiles away from exhaustion and towards increased effector function. Chronic antigen stimulation leads to CD8+ T cell exhaustion, which is mediated by persistent activation of the transcription factor NFAT in the absence of AP-1. Seo, González-Avalos and colleagues show that overexpressed BATF cooperates with IRF4 to counteract NFAT-induced exhaustion and promote better tumor control by CAR T cells in mouse models.

|
Scooped by BigField GEG Tech |

sofia carlos's curator insight,
April 10, 2022 8:44 PM
https://www.bostonterrierpetshome.com https://www.bostonterrierpetshome.com https://www.bostonterrierpetshome.com https://www.bostonterrierpetshome.com https://www.bostonterrierpetshome.com
Sign up to comment



 Your new post is loading...
Your new post is loading...
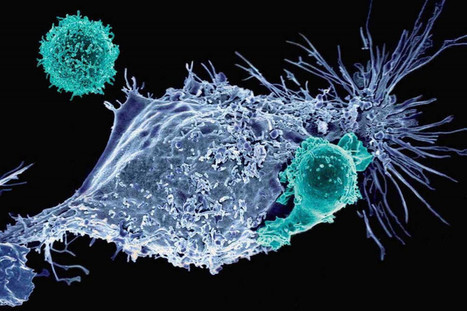






T cells that fight cancer for too long stop fighting. Researchers have named this phenomenon: T cell exhaustion. This exhaustion disrupts even the most modern cancer immunotherapies such as CAR T cells. A new study addresses this problem by giving T cells the ability to fight exhaustion itself. Researchers found that BATF, a transcription factor, cooperates with another transcription factor called IRF4 to counteract the T cell depletion program. In mouse models of melanoma and colorectal carcinoma, modifying CAR T cells to overexpress BATF by about 20-fold compared to normal cells eliminated the tumor without causing T-cell exhaustion. CAR T therapy worked against solid tumors.
Further testing showed that while IRF4 is important, it should not be overexpressed to the same degree as BATF. In addition, modified T cells also remained in place and became memory T cells. This is important because T cell depletion often prevents them from mounting a strong memory response against recurrent cancers.