 Your new post is loading...
 Your new post is loading...
Treatment with a next-generation CAR T-cell agent displayed early efficacy in a small group of patients with glioblastoma.
Over the past two decades, the immune system has attracted increasing attention for its role in fighting cancer.
A broad new strategy could hold hope for treating virtually all blood cancers with CAR T cell therapy, which is currently approved for five subtypes of blood cancer.
Decades of oncologic clinical use have demonstrated that cancer immunotherapy provides
unprecedented therapeutic benefits. Tragically, only a minority of patients respond
to existing immunotherapies. RNA lipid nanoparticles have recently emerged as modular
tools for immune stimulation. Here, we discuss advancements in RNA-based cancer immunotherapies
and opportunities for improvement.
Indiana University School of Medicine researchers have identified a new therapeutic target that could lead to more effective treatment of glaucoma.
Adding a molecular anchor to the key protein used to recognize cancer in cellular immunotherapies can make the treatments significantly more effective.
In two separate studies, researchers demonstrate how synthetic biology can be used to tackle a difficult issue in cancer immunotherapy: the way immunotherapy-related approaches focused on short-term killing of tumor cells may fail to eradicate tumors because growth of tumors happens on longer timescales.
What drives tumor growth? Is it a few rogue cells imposing their will upon healthy tissue, or diseased tissue bringing out the worst in otherwise peaceable cells? Or is it a back-and-forth, a dialogue between the two?
Researchers at VCU Massey Cancer Center have set their sights on a new therapeutic target for an aggressive form of breast cancer with limited treatment options.
The use of broad-spectrum antibiotics in the month leading up to chimeric antigen receptor T-cell therapy led to poorer outcomes and increased treatment-related toxicities, study results showed.Certain types of gut bacteria had an impact on treatment efficacy and related toxicities among patients who received CAR-T for non-Hodgkin lymphoma or acute lymphoblastic leukemia, researchers reported.
Synthetic receptor signalling has the potential to endow adoptively transferred T cells with new functions that overcome major barriers in the treatment of solid tumours, including the need for conditioning chemotherapy1,2. Here we designed chimeric receptors that have an orthogonal IL-2 receptor extracellular domain (ECD) fused with the intracellular domain (ICD) of receptors for common γ-chain (γc) cytokines IL-4, IL-7, IL-9 and IL-21 such that the orthogonal IL-2 cytokine elicits the corresponding γc cytokine signal. Of these, T cells that signal through the chimeric orthogonal IL-2Rβ-ECD–IL-9R-ICD (o9R) are distinguished by the concomitant activation of STAT1, STAT3 and STAT5 and assume characteristics of stem cell memory and effector T cells. Compared to o2R T cells, o9R T cells have superior anti-tumour efficacy in two recalcitrant syngeneic mouse solid tumour models of melanoma and pancreatic cancer and are effective even in the absence of conditioning lymphodepletion. Therefore, by repurposing IL-9R signalling using a chimeric orthogonal cytokine receptor, T cells gain new functions, and this results in improved anti-tumour activity for hard-to-treat solid tumours. Synthetic chimeric orthogonal IL-2 receptors that incorporate the intracellular domain of receptors for other γ-chain cytokines such as IL-9 can reroute orthogonal signalling and alter the phenotype of T cells to improve anti-tumour responses.
|
Magnetic resonance imaging (MRI) and lumbar puncture (LP) may not always be necessary for diagnosing and managing a serious neurological complication associated with CAR T-cell therapy, according to a new Blood Advances study.
Review synthesizes research on NK cells' role in cancer immunity and their potential in therapeutics through bioengineering, immune checkpoint inhibitors, and cell engagers, highlighting ongoing preclinical and clinical trials.
Utilizing CRISPR screening, the deubiquitinase ATXN3 has been identified as a key regulator of PD-L1 transcription in tumor cells, a critical factor in tumor immune evasion.
Immunotherapy using modified chimeric antigen receptor (CAR) T cells has greatly improved survival rates for pediatric patients with relapsed and recurrent leukemia.
Molecular routes to metastatic dissemination are critical determinants of aggressive cancers. Through in vivo CRISPR–Cas9 genome editing, we generated somatic mosaic genetically engineered models that faithfully recapitulate metastatic renal tumors. Disruption of 9p21 locus is an evolutionary driver to systemic disease through the rapid acquisition of complex karyotypes in cancer cells. Cross-species analysis revealed that recurrent patterns of copy number variations, including 21q loss and dysregulation of the interferon pathway, are major drivers of metastatic potential. In vitro and in vivo genomic engineering, leveraging loss-of-function studies, along with a model of partial trisomy of chromosome 21q, demonstrated a dosage-dependent effect of the interferon receptor genes cluster as an adaptive mechanism to deleterious chromosomal instability in metastatic progression. This work provides critical knowledge on drivers of renal cell carcinoma progression and defines the primary role of interferon signaling in constraining the propagation of aneuploid clones in cancer evolution. Using genetically engineered models, Genovese and colleagues study patterns of convergent evolution in renal cancer, and pinpoint dysregulation of interferon signaling as a means of adaptation to chromosomal instability in metastatic progression.
A new approach that delivers a "one-two punch" to help T cells attack solid tumors is the focus of a preclinical study by researchers from the Perelman School of Medicine at the University of Pennsylvania.
Pancreatic cancer is an incurable form of cancer, and gene therapies are currently in clinical testing to treat this deadly disease. A comprehensive review of the gene and cell biotherapies in development to combat pancreatic cancer is published in the peer-reviewed journal Human Gene Therapy.
The recent years have seen a wave of adoptive cell therapies (ACTs), a type of immunotherapy in which T cells (T cell transfer therapy) and other immune cells are obtained from patients, activated and multiplied outside the body, and infused in larger numbers back into the blood circulation to help fight cancers.
Precision-controlled CAR-T-cell immunotherapies could be used to tackle a range of tumour types.
Last week, PACT Pharma shared results from the first clinical trial using CRISPR to direct patients’ immune cells to treat solid tumours. The findings, which were published in an unedited manuscript in Nature, provide early proof-of-concept that patient immune cells can be reprogrammed to attack their own cancer. The results were als
One of the greatest challenges facing cancer researchers is to understand why some patients don't respond to treatments.
Scientists at St. Jude Children's Research Hospital identified a molecular mechanism that in a preclinical study unlocked the promise of CAR T–cell therapy for treatment of solid tumors.
|



 Your new post is loading...
Your new post is loading...

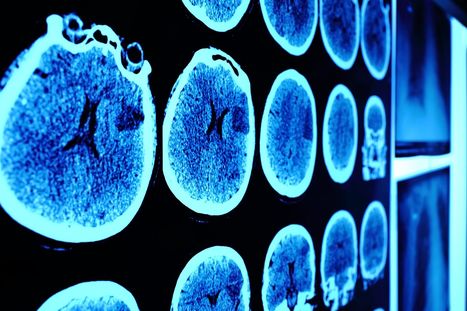


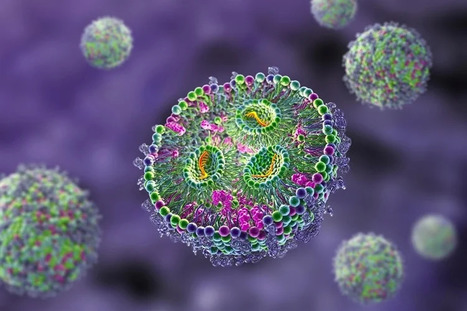

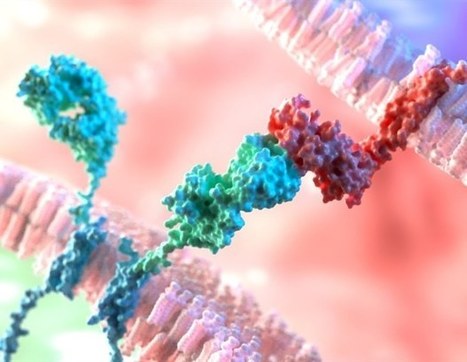
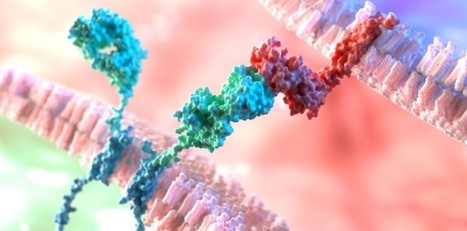










CARv3-TEAM-E T cells are CAR T cells targeting EGFR variant III tumor-specific antigen (EGFRvIII) in addition to wild-type EGFR. According to the results of the phase 1 INCIPIENT trial (NCT05660369) published in the New England Journal of Medicine, preliminary results in humans demonstrated that all 3 glioblastoma patients treated with CARv3-TEAM-E T cells between March 2023 and July 2023 showed dramatic and rapid radiographic regression of their tumors within days of receiving CARv3-TEAM-E T cells via a single intraventricular infusion. Responses were transient in 2 of the patients, however 1 patient, a 72-year-old man, showed an 18.5% decrease in tumor cross-sectional area on day 2 after receiving a single infusion of 10 x 106 CAR-positive CARv3-TEAM-E T cells. Moreover, 69 days after infusion, tumor cross-sectional area had decreased by a further 60.7% from baseline; the response was sustained and continued to improve at the last assessment, which took place more than 150 days after infusion.