 Your new post is loading...
 Your new post is loading...
This study is led by Prof. Xianqun Fan (Department of Ophthalmology, Shanghai Jiao Tong University School of Medicine Affiliated Ninth People's Hospital).
Utilizing CRISPR screening, the deubiquitinase ATXN3 has been identified as a key regulator of PD-L1 transcription in tumor cells, a critical factor in tumor immune evasion.
Rochester Institute of Technology researchers are improving non-invasive treatment options for degenerative disc disease, an ailment that impacts 3 million adults yearly in the U.S., according to the Mayo Clinic.
Induced pluripotent stem (iPS) cells have a great impact on biology and medicine, and they are expected to improve regenerative medicine.
Chimeric antigen receptors (CARs) and synthetic Notch (synNotch) receptors are engineered cell-surface receptors that sense a target antigen and respond by activating T cell receptor signaling or a customized gene program, respectively. Here, to expand the targeting capabilities of these receptors, we develop “universal” receptor systems for which receptor specificity can be directed post-translationally via covalent attachment of a co-administered antibody bearing a benzylguanine (BG) motif. A SNAPtag self-labeling enzyme is genetically fused to the receptor and reacts with BG-conjugated antibodies for covalent assembly, programming antigen recognition. We demonstrate that activation of SNAP-CAR and SNAP-synNotch receptors can be successfully targeted by clinically relevant BG-conjugated antibodies, including anti-tumor activity of SNAP-CAR T cells in vivo in a human tumor xenograft mouse model. Finally, we develop a mathematical model to better define the parameters affecting universal receptor signaling. SNAP receptors provide a powerful strategy to post-translationally reprogram the targeting specificity of engineered cells. Chimeric antigen receptors (CARs) and synthetic Notch (synNotch) receptors are promising platforms for cell-based immunotherapies. Here, the authors develop highly programmable versions of these receptors that can be universally targeted to antigens of interest through covalent enzyme chemistry.
A new approach that delivers a "one-two punch" to help T cells attack solid tumors is the focus of a preclinical study by researchers from the Perelman School of Medicine at the University of Pennsylvania.
Adding a molecular anchor to the key protein used to recognize cancer in cellular immunotherapies can make the treatments significantly more effective.
The recent years have seen a wave of adoptive cell therapies (ACTs), a type of immunotherapy in which T cells (T cell transfer therapy) and other immune cells are obtained from patients, activated and multiplied outside the body, and infused in larger numbers back into the blood circulation to help fight cancers.
A new approach to cancer immunotherapy that uses one type of immune cell to kill another-;rather than directly attacking the cancer-;provokes a robust anti-tumor immune response that shrinks ovarian, lung, and pancreatic tumors in preclinical disease models, according to researchers at the Icahn School of Medicine at Mount Sinai in New York.
A study published this month in the New England Journal of Medicine (NEJM) may change the standard of care for adults who relapse from large B-cell lymphoma.
Continuous antigen exposure drives CAR T cell exhaustion and promotes CD8+ T-to-NK-like
T cell transition. Transcription factors ID3 and SOX4 are upregulated during CAR dysfunction
and regulate genes associated with exhaustion, including NK receptors. Knocking out
ID3 and SOX4 in CAR T cells slows dysfunction and improves anti-tumor immunity.
Chimeric antigen receptor T-cell therapy followed by pembrolizumab appeared safe and feasible for patients with malignant pleural mesothelioma, phase 1 trial results published in Cancer Discovery showed.Eighty-three percent of patients who received the regimen remained alive 1 year after CAR-T infusion. Researchers reported no cases of high-grade cytokine release syndrome or neurotoxicity.
|
At EPFL's School of Engineering, Professor Li Tang's Laboratory of Biomaterials for Immunoengineering has made significant strides in cancer treatment research.
Immunotherapy using modified chimeric antigen receptor (CAR) T cells has greatly improved survival rates for pediatric patients with relapsed and recurrent leukemia.
In the context of relapsed and refractory childhood pre-B cell acute lymphoblastic leukemia (R/R B-ALL), CD19-targeting chimeric antigen receptor (CAR)-T cells often induce durable remissions, which requires the persistence of CAR-T cells. In this study, we systematically analyzed CD19 CAR-T cells of 10 children with R/R B-ALL enrolled in the CARPALL trial via high-throughput single-cell gene expression and T cell receptor sequencing of infusion products and serial blood and bone marrow samples up to 5 years after infusion. We show that long-lived CAR-T cells developed a CD4/CD8 double-negative phenotype with an exhausted-like memory state and distinct transcriptional signature. This persistence signature was dominant among circulating CAR-T cells in all children with a long-lived treatment response for which sequencing data were sufficient (4/4, 100%). The signature was also present across T cell subsets and clonotypes, indicating that persisting CAR-T cells converge transcriptionally. This persistence signature was also detected in two adult patients with chronic lymphocytic leukemia with decade-long remissions who received a different CD19 CAR-T cell product. Examination of single T cell transcriptomes from a wide range of healthy and diseased tissues across children and adults indicated that the persistence signature may be specific to long-lived CAR-T cells. These findings raise the possibility that a universal transcriptional signature of clinically effective, persistent CD19 CAR-T cells exists. In children with relapsed or refractory B cell acute lymphoblastic leukemia and in complete remission after CD19 CAR-T cell therapy, long-lived CAR-T cells express a persistence gene signature that is also present in persistent CD19 CAR-T cells from adults with chronic lymphocytic leukemia.
Vα24-invariant natural killer T cells (NKTs) have anti-tumor properties that can be enhanced by chimeric antigen receptors (CARs). Here we report updated interim results from the first-in-human phase 1 evaluation of autologous NKTs co-expressing a GD2-specific CAR with interleukin 15 (IL15) (GD2-CAR.15) in 12 children with neuroblastoma (NB). The primary objectives were safety and determination of maximum tolerated dose (MTD). The anti-tumor activity of GD2-CAR.15 NKTs was assessed as a secondary objective. Immune response evaluation was an additional objective. No dose-limiting toxicities occurred; one patient experienced grade 2 cytokine release syndrome that was resolved by tocilizumab. The MTD was not reached. The objective response rate was 25% (3/12), including two partial responses and one complete response. The frequency of CD62L+NKTs in products correlated with CAR-NKT expansion in patients and was higher in responders (n = 5; objective response or stable disease with reduction in tumor burden) than non-responders (n = 7). BTG1 (BTG anti-proliferation factor 1) expression was upregulated in peripheral GD2-CAR.15 NKTs and is a key driver of hyporesponsiveness in exhausted NKT and T cells. GD2-CAR.15 NKTs with BTG1 knockdown eliminated metastatic NB in a mouse model. We conclude that GD2-CAR.15 NKTs are safe and can mediate objective responses in patients with NB. Additionally, their anti-tumor activity may be enhanced by targeting BTG1. ClinicalTrials.gov registration: NCT03294954 . In updated results from a phase 1 trial of GD2-specific CAR-NKT cells in patients with neuroblastoma, no dose-limiting toxicities were observed across multiple dose levels; the maximum tolerated dose was not reached; and there was evidence of anti-tumor activity.
Chimeric antigen receptor T-cell (CAR-T) therapy has transformed cancer treatment, yet relatively few studies have investigated the impact of the therapy on longitudinal patient quality of life – an aspect of care that often suffers from receiving traditional intensive cancer medications, such as chemotherapy.
Indiana University School of Medicine researchers have identified a new therapeutic target that could lead to more effective treatment of glaucoma.
With a slew of tools to trick out immune cells, researchers are expanding the repertoire of CAR-T therapies.
Researchers at Great Ormond Street Hospital for Children (GOSH) and UCL Great Ormond Street Institute of Child Health (UCL GOS ICH) have used CRISPR/Cas9 technology to engineer donor T cells to try to treat seriously ill children with resistant leukaemia, who had otherwise exhausted all available therapies.
Scientists at St. Jude Children's Research Hospital identified a molecular mechanism that in a preclinical study unlocked the promise of CAR T–cell therapy for treatment of solid tumors.
B cell-activating factor (BAFF) binds the three receptors BAFF-R, BCMA, and TACI, predominantly expressed on mature B cells. Almost all B cell cancers are reported to express at least one of these receptors. Here we develop a BAFF ligand-based chimeric antigen receptor (CAR) and generate BAFF CAR-T cells using a non-viral gene delivery method. We show that BAFF CAR-T cells bind specifically to each of the three BAFF receptors and are effective at killing multiple B cell cancers, including mantle cell lymphoma (MCL), multiple myeloma (MM), and acute lymphoblastic leukemia (ALL), in vitro and in vivo using different xenograft models. Co-culture of BAFF CAR-T cells with these tumor cells results in induction of activation marker CD69, degranulation marker CD107a, and multiple proinflammatory cytokines. In summary, we report a ligand-based BAFF CAR-T capable of binding three different receptors, minimizing the potential for antigen escape in the treatment of B cell cancers. Antigen escape represents a potential drawback of chimeric antigen receptor T cell (CAR-T) therapy targeting a single tumor-associated antigen. To reduce the risk of antigen escape, here the authors report the design and characterization of a BAFF ligand CAR-T that can recognize three different receptors (BAFF-R, BCMA and TACI), demonstrating in vitro and in vivo cytotoxicity against multiple B cell cancer models.
Mount Sinai scientists have become the first to report a potentially serious side effect related to a new form of immunotherapy known as CAR-T cell therapy, which was recently approved for the treatment of multiple myeloma.
In this study, Jalil and colleagues develop and test a method for simultaneous hiPSC
reprogramming and RNA-delivered adenine base editing. Starting from primary patient-derived
fibroblasts, this approach yields several on-target edited isogenic hiPSC lines, with
remarkable speed and editing efficiency and without compromising their genetic integrity
or their pluripotency quality. The gene editing corrects the cellular disease phenotype.
|



 Your new post is loading...
Your new post is loading...




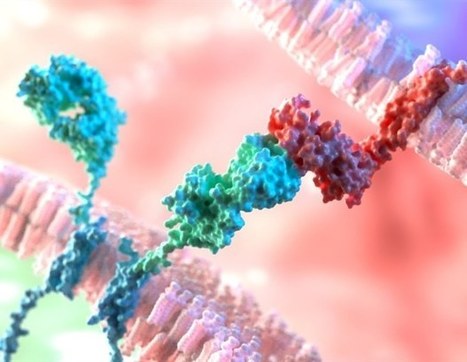
















In a review, researchers detail the development history of Principal Editing (PE), the latest evolution of CRISPR-Cas-based technologies. PE was proposed by a team of researchers in 2019, which is characterized by the absence of double-strand breaks (DSBs) or homology sequence patterns with variable application scenarios, including point mutations as well as insertions or deletions. The PE system consists of two parts: the master editors (PEs) and the master editing guide RNA (pegRNA). This PE system has developed and progressed rapidly over the last four years, with versatile advances in its architecture to increase editing efficiency, targeting and specificity, including a new pegRNA design, PE modification and improved delivery. Moreover, despite its relatively recent inception, PE has been widely applied to correct pathological mutations associated with genetic diseases, both in vitro and in vivo , presenting great potential for advancing the field of gene therapy from bench to bedside.