 Your new post is loading...
 Your new post is loading...
Targeting two brain tumor-associated proteins-;rather than one-;with CAR T cell therapy shows promise as a strategy for reducing solid tumor growth in patients with recurrent glioblastoma (GBM), an aggressive form of brain cancer, according to early results from the first six patients treated in an ongoing Phase I clinical trial led by researchers from the Perelman School of Medicine at the University of Pennsylvania and Penn Medicine's Abramson Cancer Center.
This study is led by Prof. Xianqun Fan (Department of Ophthalmology, Shanghai Jiao Tong University School of Medicine Affiliated Ninth People's Hospital).
Using CRISPR, an immune system bacteria use to protect themselves from viruses, scientists have harnessed the power to edit genetic information within cells.
At EPFL's School of Engineering, Professor Li Tang's Laboratory of Biomaterials for Immunoengineering has made significant strides in cancer treatment research.
The recent publication in Science by Mogila, Tamulaitiene et al. represents a continuation of the successful scientific research conducted by Gintautas Tamulaitis' group.
Immunotherapy using modified chimeric antigen receptor (CAR) T cells has greatly improved survival rates for pediatric patients with relapsed and recurrent leukemia.
The gene editing technique CRISPR/Cas9 has allowed researchers to make precise and impactful changes to an organism's DNA to fix mutations that cause genetic disease.
Gene therapy that alters hemoglobin genes may be an answer to curing sickle cell disease (SCD) and beta thalassemia. These two common life-threatening anemias afflict millions of individuals across the globe. Scientists at St. Jude Children's Research Hospital and the Broad Institute of MIT and Harvard used a next-generation genome editing technology, adenosine base editing, to restart fetal hemoglobin expression in SCD patient cells. The approach raised the expression of fetal hemoglobin to higher, more stable, and more uniform levels than other genome editing technologies that use CRISPR/Cas9 nuclease in human hematopoietic stem cells. The findings were published in Nature Genetics.
The expanding applications of nonviral genomic medicines in the lung remain restricted by delivery challenges. Here, leveraging a high-throughput platform, we synthesize and screen a combinatorial library of biodegradable ionizable lipids to build inhalable delivery vehicles for messenger RNA and CRISPR–Cas9 gene editors. Lead lipid nanoparticles are amenable for repeated intratracheal dosing and could achieve efficient gene editing in lung epithelium, providing avenues for gene therapy of congenital lung diseases. A high-throughput screen improves lipid nanoparticle delivery of gene editors in the lung.
A new approach that delivers a "one-two punch" to help T cells attack solid tumors is the focus of a preclinical study by researchers from the Perelman School of Medicine at the University of Pennsylvania.
Range of DNA repair in response to double-strand breaks induced in human preimplantation embryos remains uncertain due to the complexity of analyzing single- or few-cell samples. Sequencing of such minute DNA input requires a whole genome amplification that can introduce artifacts, including coverage nonuniformity, amplification biases, and allelic dropouts at the target site. We show here that, on average, 26.6% of preexisting heterozygous loci in control single blastomere samples appear as homozygous after whole genome amplification indicative of allelic dropouts. To overcome these limitations, we validate on-target modifications seen in gene edited human embryos in embryonic stem cells. We show that, in addition to frequent indel mutations, biallelic double-strand breaks can also produce large deletions at the target site. Moreover, some embryonic stem cells show copy-neutral loss of heterozygosity at the cleavage site which is likely caused by interallelic gene conversion. However, the frequency of loss of heterozygosity in embryonic stem cells is lower than in blastomeres, suggesting that allelic dropouts is a common whole genome amplification outcome limiting genotyping accuracy in human preimplantation embryos. DNA repair in response to DSBs in the preimplantation embryo is hard to analyze. Here the authors show that over 25% of pre-existing heterozygous loci in control single blastomere samples appeared as homozygous after whole genome amplification, therefore, they validated gene editing seen in human embryos in ESCs.
Understanding moral acceptability and willingness to use is crucial for informing policy
|
Current approaches for inserting autonomous transgenes into the genome, such as CRISPR–Cas9 or virus-based strategies, have limitations including low efficiency and high risk of untargeted genome mutagenesis. Here, we describe precise RNA-mediated insertion of transgenes (PRINT), an approach for site-specifically primed reverse transcription that directs transgene synthesis directly into the genome at a multicopy safe-harbor locus. PRINT uses delivery of two in vitro transcribed RNAs: messenger RNA encoding avian R2 retroelement-protein and template RNA encoding a transgene of length validated up to 4 kb. The R2 protein coordinately recognizes the target site, nicks one strand at a precise location and primes complementary DNA synthesis for stable transgene insertion. With a cultured human primary cell line, over 50% of cells can gain several 2 kb transgenes, of which more than 50% are full-length. PRINT advantages include no extragenomic DNA, limiting risk of deleterious mutagenesis and innate immune responses, and the relatively low cost, rapid production and scalability of RNA-only delivery. Transgenes are inserted into human cells by 2-RNA delivery of a retroelement protein and template.
Chimeric antigen receptor T-cell therapy, or CAR T, has dramatically improved the treatment of certain blood cancers. Initially approved for patients who had failed multiple lines of therapy, clinical trials have shown CAR T can be used as an earlier treatment option.
Researchers developed an RNA-based switch, the pA regulator system, to control gene expression in mammalian cells by modulating synthetic polyA signal cleavage, offering a novel approach for gene therapy applications.
Utilizing CRISPR screening, the deubiquitinase ATXN3 has been identified as a key regulator of PD-L1 transcription in tumor cells, a critical factor in tumor immune evasion.
The gene-editing technology CRISPR shows early promise as a therapeutic strategy for the aggressive and difficult-to-treat brain cancer known as primary glioblastoma, according to findings of a new study from Gladstone Institutes.
RAG2-SCID is a primary immunodeficiency caused by mutations in Recombination-activating gene 2 (RAG2), a gene intimately involved in the process of lymphocyte maturation and function. ex-vivo manipulation of a patient’s own hematopoietic stem and progenitor cells (HSPCs) using CRISPR-Cas9/rAAV6 gene editing could provide a therapeutic alternative to the only current treatment, allogeneic hematopoietic stem cell transplantation (HSCT). Here we show an innovative RAG2 correction strategy that replaces the entire endogenous coding sequence (CDS) for the purpose of preserving the critical endogenous spatiotemporal gene regulation and locus architecture. Expression of the corrective transgene leads to successful development into CD3+TCRαβ+ and CD3+TCRγδ+ T cells and promotes the establishment of highly diverse TRB and TRG repertoires in an in-vitro T-cell differentiation platform. Thus, our proof-of-concept study holds promise for safer gene therapy techniques of tightly regulated genes. RAG2-SCID is a primary immunodeficiency caused by mutations in Recombination-activating gene 2 (RAG2). Here the authors report a RAG2 correction strategy that replaces the entire endogenous coding sequence (CDS) to maintain the endogenous spatiotemporal gene regulation and locus architecture.
Induced pluripotent stem (iPS) cells have a great impact on biology and medicine, and they are expected to improve regenerative medicine.
Vα24-invariant natural killer T cells (NKTs) have anti-tumor properties that can be enhanced by chimeric antigen receptors (CARs). Here we report updated interim results from the first-in-human phase 1 evaluation of autologous NKTs co-expressing a GD2-specific CAR with interleukin 15 (IL15) (GD2-CAR.15) in 12 children with neuroblastoma (NB). The primary objectives were safety and determination of maximum tolerated dose (MTD). The anti-tumor activity of GD2-CAR.15 NKTs was assessed as a secondary objective. Immune response evaluation was an additional objective. No dose-limiting toxicities occurred; one patient experienced grade 2 cytokine release syndrome that was resolved by tocilizumab. The MTD was not reached. The objective response rate was 25% (3/12), including two partial responses and one complete response. The frequency of CD62L+NKTs in products correlated with CAR-NKT expansion in patients and was higher in responders (n = 5; objective response or stable disease with reduction in tumor burden) than non-responders (n = 7). BTG1 (BTG anti-proliferation factor 1) expression was upregulated in peripheral GD2-CAR.15 NKTs and is a key driver of hyporesponsiveness in exhausted NKT and T cells. GD2-CAR.15 NKTs with BTG1 knockdown eliminated metastatic NB in a mouse model. We conclude that GD2-CAR.15 NKTs are safe and can mediate objective responses in patients with NB. Additionally, their anti-tumor activity may be enhanced by targeting BTG1. ClinicalTrials.gov registration: NCT03294954 . In updated results from a phase 1 trial of GD2-specific CAR-NKT cells in patients with neuroblastoma, no dose-limiting toxicities were observed across multiple dose levels; the maximum tolerated dose was not reached; and there was evidence of anti-tumor activity.
CRISPR-Cas9 is widely used to edit the genome by studying genes of interest and modifying disease-associated genes.
Chimeric antigen receptor T-cell (CAR-T) therapy has transformed cancer treatment, yet relatively few studies have investigated the impact of the therapy on longitudinal patient quality of life – an aspect of care that often suffers from receiving traditional intensive cancer medications, such as chemotherapy.
Indiana University School of Medicine researchers have identified a new therapeutic target that could lead to more effective treatment of glaucoma.
Pancreatic cancer is an incurable form of cancer, and gene therapies are currently in clinical testing to treat this deadly disease. A comprehensive review of the gene and cell biotherapies in development to combat pancreatic cancer is published in the peer-reviewed journal Human Gene Therapy.
|



 Your new post is loading...
Your new post is loading...

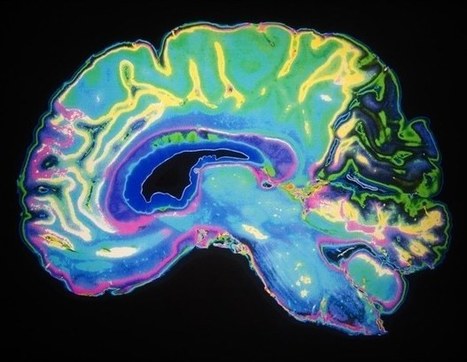





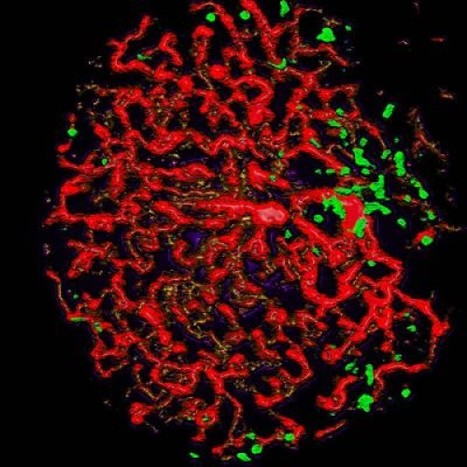











Glioblastoma (GBM) is the most common and aggressive type of cancerous brain tumor in adults. People with GBM generally expect to live 12 to 18 months after diagnosis. Despite decades of research, there is no known cure for GBM, and treatments have only a limited effect on extending an individual's life expectancy. However, researchers have tested a technology that delivers CAR-T cells targeting two proteins commonly found in brain tumors: epidermal growth factor receptor (EGFR), estimated to be present in 60% of all GBMs, and interleukin-13 receptor alpha 2 (IL13Rα2), which is expressed in over 75% of GBMs. While CAR-T cell therapy for blood cancers is usually administered intravenously, the researchers administered these dual-targeted CAR-T cells intrathecally, by injection into the cerebrospinal fluid, so that the modified cells could reach the tumors more directly in the brain. Magnetic Resonance Imaging scans taken 24 to 48 hours after administration of dual-targeted CAR-T cells targeting EGFR and IL13Rα2 revealed a reduction in tumor size in all six patients, and these reductions were maintained up to several months later in a subgroup of patients.