Despite their clinical success, chimeric antigen receptor (CAR)-T cell therapies for B cell malignancies are limited by lengthy, costly and labor-intensive ex vivo manufacturing procedures that might lead to cell products with heterogeneous composition. Here we describe an implantable Multifunctional Alginate Scaffold for T Cell Engineering and Release (MASTER) that streamlines in vivo CAR-T cell manufacturing and reduces processing time to a single day. When seeded with human peripheral blood mononuclear cells and CD19-encoding retroviral particles, MASTER provides the appropriate interface for viral vector-mediated gene transfer and, after subcutaneous implantation, mediates the release of functional CAR-T cells in mice. We further demonstrate that in vivo-generated CAR-T cells enter the bloodstream and control distal tumor growth in a mouse xenograft model of lymphoma, showing greater persistence than conventional CAR-T cells. MASTER promises to transform CAR-T cell therapy by fast-tracking manufacture and potentially reducing the complexity and resources needed for provision of this type of therapy. Implantable scaffolds rapidly generate and release anti-tumor CAR-T cells in mice.

|
Scooped by BigField GEG Tech |

sofia carlos's curator insight,
April 10, 2022 8:40 PM
https://www.bostonterrierpetshome.com
Sign up to comment



 Your new post is loading...
Your new post is loading...
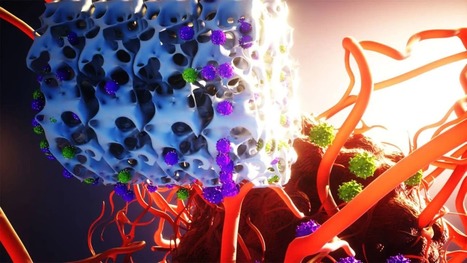






Many people are excluded from CAR T cell-based treatments because of its cost. One reason for the high cost is that the manufacturing process is complex, time-consuming and must be individually tailored to each cancer patient. So to address this challenge, the researchers created a biotechnology called Multifunctional Alginate Scaffolds for T cell Engineering and Release (MASTER) that is a biocompatible sponge-like material. To begin treatment, the researchers isolate the patient's T cells and mix these naive T cells with the modified virus. The researchers pour this mixture onto MASTER, which absorbs it. MASTER is decorated with the antibodies that activate the T cells, so the cell activation process begins almost immediately. Meanwhile, MASTER is surgically implanted into the patient. After implantation, as the T cells become activated, they begin to respond to the modified viruses, which reprogram them into CAR-T cells. MASTER is also imbued with interleukin factors that promote cell proliferation. After implantation, these interleukins begin to leach out, promoting rapid proliferation of CAR-T cells. In a proof-of-concept study involving lymphoma in mice, researchers found that this treatment was faster and more effective than conventional CAR-T cell cancer treatment.