The lack of registered drugs for nonalcoholic fatty liver disease (NAFLD) is partly due to the paucity of human-relevant models for target discovery and compound screening. Here we use human fetal hepatocyte organoids to model the first stage of NAFLD, steatosis, representing three different triggers: free fatty acid loading, interindividual genetic variability (PNPLA3 I148M) and monogenic lipid disorders (APOB and MTTP mutations). Screening of drug candidates revealed compounds effective at resolving steatosis. Mechanistic evaluation of effective drugs uncovered repression of de novo lipogenesis as the convergent molecular pathway. We present FatTracer, a CRISPR screening platform to identify steatosis modulators and putative targets using APOB−/− and MTTP−/− organoids. From a screen targeting 35 genes implicated in lipid metabolism and/or NAFLD risk, FADS2 (fatty acid desaturase 2) emerged as an important determinant of hepatic steatosis. Enhancement of FADS2 expression increases polyunsaturated fatty acid abundancy which, in turn, reduces de novo lipogenesis. These organoid models facilitate study of steatosis etiology and drug targets. Organoid models of early liver disease aid target discovery and drug screening.
Research and publish the best content.
Get Started for FREE
Sign up with Facebook Sign up with X
I don't have a Facebook or a X account
Already have an account: Login
 Your new post is loading... Your new post is loading...
 Your new post is loading... Your new post is loading...
|
|




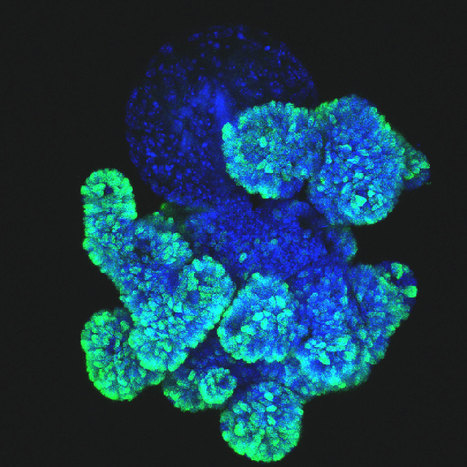







The accumulation of fat in the liver is an increasingly common disease worldwide, with more than a quarter of the world's population affected. Various causes can lead to the development of fatty liver, with diet and lifestyle being the most common contributors. There is currently no treatment for fatty liver that can stop or reverse the disease. Therefore, researchers have established new human organoid models of fatty liver. They used these models to shed light on drug responses and established a CRISPR screening platform, called FatTracer, to identify new disease mediators and potential therapeutic targets. After screening 35 candidates, a critical new role for the FADS2 (fatty acid desaturase 2) gene in fatty liver disease was discovered. Disruption of FADS2 made the organoids much fatter. Upon overexpression of FADS2, the hepatic steatosis that the organoids once displayed was severely reduced. These models will therefore help test and develop new drugs to treat hepatic steatosis and better understand the biology of the disease. The results of the study will be published in Nature Biotechnology.