Research and publish the best content.
Get Started for FREE
Sign up with Facebook Sign up with X
I don't have a Facebook or a X account
Already have an account: Login
 Your new post is loading... Your new post is loading...
 Your new post is loading... Your new post is loading...
|
|




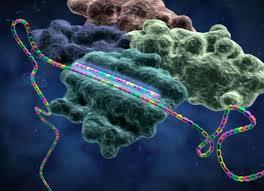










Challenges in the delivery of small interfering RNA (siRNA) have hampered clinical translation. Polymeric or periodic short hairpin RNAs (p-shRNAs) are a recent development that can potentially address these delivery barriers by showing improved stability and complexation to enable nanoparticle packaging. Here The scientists modify these biomacromolecules packaging via structural and sequence engineering coupled with selective enzymatic digestion to generate an open-ended p-shRNA (op-shRNA) that is cleaved over ten times more efficiently to yield siRNA. The op-shRNA induces considerably greater gene silencing than p-shRNA in multiple cancer cell lines up to nine days. Op-shRNA provides an RNAi platform that can potentially be packaged and delivered efficiently to disease sites with higher therapeutic efficacy.