Siteman Cancer Center, based at Barnes-Jewish Hospital and Washington University School of Medicine in St. Louis, is one of the first centers nationwide to offer a newly approved cell-based immunotherapy that targets melanoma.
Research and publish the best content.
Get Started for FREE
Sign up with Facebook Sign up with X
I don't have a Facebook or a X account
Already have an account: Login
 Your new post is loading... Your new post is loading...
 Your new post is loading... Your new post is loading...
|




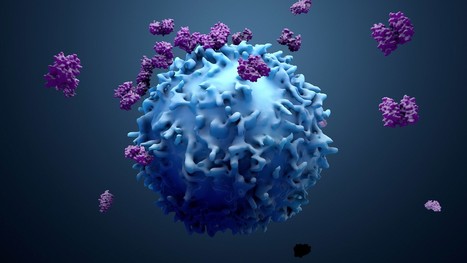






Following the approval from the Food and Drug Administration (FDA), doctors at the University of Washington's Siteman Cancer Center will administer tumor-infiltrating lymphocyte (TIL) therapy to treat certain adult patients with metastatic melanoma, an aggressive skin cancer that has spread to other areas of the body. The treatment is intended for patients with metastatic melanoma that cannot be treated by surgery and has continued to grow and spread despite having already been heavily treated with other approved strategies, including chemotherapy and immune checkpoint inhibitors. The first centers to administer TIL therapy are those with extensive expertise in treating patients with cellular immunotherapies, such as CAR-T cell therapy for blood cancers. For the therapy, doctors at an approved treatment center take a sample of the tumor and send the tissue to an Iovance manufacturing facility, where tumor-infiltrating lymphocytes are isolated from the tumor and then expanded outside the body. This TIL therapy cell product is then cryopreserved and returned to the patient. When returned to the patient's body via intravenous infusion, the tumor-specific T cells, now numbering in the billions, are much more effective at killing tumor cells throughout the body.