Actinium Pharma gets rights to develop & commercialize BC8 monoclonal antibody from FHCRC (Actinium Pharma gets rights to develop & commercialize BC8 monoclonal antibody from FHCRC http://t.co/7ZIExyFe...)...
Get Started for FREE
Sign up with Facebook Sign up with X
I don't have a Facebook or a X account


 Your new post is loading... Your new post is loading...
 Your new post is loading... Your new post is loading...
|

|
Scooped by Gilbert C FAURE |
Le 24 mai, des chercheurs chinois ont annoncé avoir transplanté avec succès un foie de porc génétiquement modifié sur un patient humain vivant atteint d’un cancer du foie.

|
Scooped by Gilbert C FAURE |
A l'Institut de recherche sur les dispositifs médicaux de Suzhou de l'Université du Sud-Est, situé à Suzhou, dans la province chinoise du Jiangsu (est), on a...

|
Scooped by Gilbert C FAURE |
SignificanceRespiratory virus infections in humans are a significant global health concern, causing a wide range of diseases with substantial morbidity and mortality worldwide. This underscores the...

|
Scooped by Gilbert C FAURE |
Aspirin, taken by around 29 million Americans daily, has increasingly been linked to inhibiting the growth of certain cancers – but we didn't quite know how. Now, scientists have uncovered how it helps the immune system see and kill cancer cells.

|
Scooped by Gilbert C FAURE |
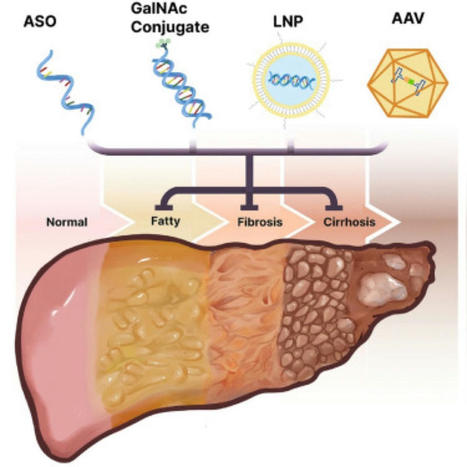
“#Oligonucleotide Therapies for Nonalcoholic #Steatohepatitis”
Cell Press link: https://lnkd.in/eWwMNyCw

|
Scooped by Gilbert C FAURE |
Biotechnology firm LyGenesis has injected donor cells into a person with liver failure for the first time.

|
Scooped by Gilbert C FAURE |
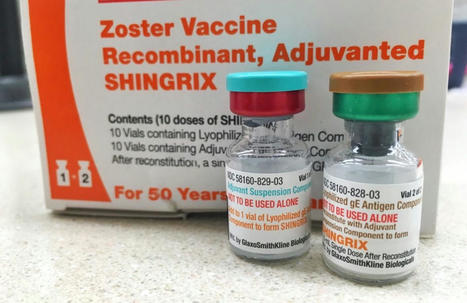
Protection results from GSK Shingrix vaccines against #Shingles are impressive. Guess which adjuvant was used?
👉A #TLR4 agonist of course 🦠
The results…

|
Scooped by Gilbert C FAURE |
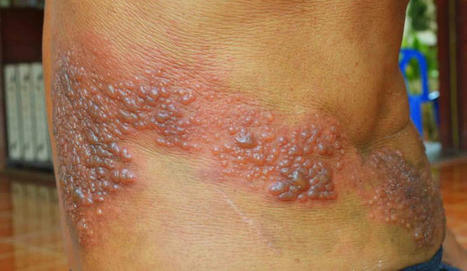
🔸 Actualisation de la stratégie vaccinale contre le zona :
Le zona, causé par la réactivation du virus varicelle-zona, est une maladie virale touchant…

|
Scooped by Gilbert C FAURE |
"The modulation of cytokines and cytokine receptors is highly promising for enhancing NK cell survival and persistence, thus paving the way for durable…

|
Scooped by Gilbert C FAURE |
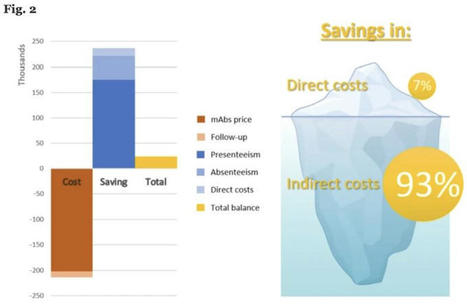
Bravo à nos collègues de Barcelone qui ont démontré le bénéfice medico-économique du remboursement des anti-CGRP en Espagne, notamment grâce à la réduction de…

|
Scooped by Gilbert C FAURE |
OverviewOn 22 February 2024, the Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion, recommending the granting of a marketing authorisation for the medicinal product Tizveni, intended for the treatment of locally advanced or metastatic non-small cell lung cancer (NSCLC) either in monotherapy or in combination with chemotherapy. The applicant for this medicinal product is Beigene Ireland Limited.Tizveni will be available as a 100 mg concentrate for solution for infusion. The active substance of Tizveni is tislelizumab, an antineoplastic agent (ATC code: L01FF09). Tislelizumab is a humanised IgG4 variant monoclonal antibody that potentiates T-cell responses, including anti-tumour responses, through blockade of PD-1 binding to PD-L1 and PD-L2 ligands.The benefit of Tizveni is an improvement in overall survival and progression free survival in patients with locally advanced or metastatic NSCLC, as shown in three open-label, randomised phase 3 studies comparing Tizveni (either in monotherapy or in combination) with chemotherapy. The most common side effects are anaemia, fatigue and increased AST.The full indication is:Tizveni in combination with pemetrexed and platinum‑containing chemotherapy is indicated for the first-line treatment of adult patients with non-squamous non-small cell lung cancer whose tumours have PD-L1 expression on ≥50% of tumour cells with no EGFR or ALK positive mutations and who have:locally advanced NSCLC and are not candidates for surgical resection or platinum-based chemoradiation, ormetastatic NSCLC.Tizveni in combination with carboplatin and either paclitaxel or nab-paclitaxel is indicated for the first-line treatment of adult patients with squamous non-small cell lung cancer who have:locally advanced NSCLC and are not candidates for surgical resection or platinum-based chemoradiation, ormetastatic NSCLC.Tizveni as monotherapy is indicated for the treatment of adult patients with locally advanced or metastatic non-small cell lung cancer after prior platinum-based therapy. Patients with EGFR mutant or ALK positive NSCLC should also have received targeted therapies before receiving tislelizumab.Tizveni should be prescribed by physicians experienced in the treatment of cancer.Detailed recommendations for the use of this product will be described in the summary of product characteristics (SmPC), which will be published in the European public assessment report (EPAR) and made available in all official European Union languages after the marketing authorisation has been granted by the European Commission.CHMP summary of positive opinion for TizveniAdoptedFirst published: 23/02/2024Reference Number: EMA/CHMP/59203/2024 English (EN) (114.24 KB - PDF)ViewProduct detailsName of medicine Tizveni Active substance Tislelizumab International non-proprietary name (INN) or common name tislelizumab Therapeutic area (MeSH) Carcinoma, Non-Small-Cell Lung Anatomical therapeutic chemical (ATC) code L01FF09 EMA product number EMEA/H/C/005542 Marketing authorisation applicant BeiGene Ireland Ltd Opinion adopted 22/02/2024 Opinion status Positive This page was last updated on 23/02/2024Share this page

|
Scooped by Gilbert C FAURE |
There are other ways to therapeutically stimulate intratumoral innate immunity - with encouraging clinical results -, including in situ vaccination against the…

|
Scooped by Gilbert C FAURE |

|
Scooped by Gilbert C FAURE |
Targeting claudin-1 offers a promising approach for treating solid tumours by selectively killing cancer cells and breaking down barriers to immunotherapy.

|
Scooped by Gilbert C FAURE |
Antibody–drug conjugates have emerged as a powerful tool to deliver highly targeted cancer therapies. Next-generation antibody-drug conjugate designs aim to push the envelope even further.

|
Scooped by Gilbert C FAURE |
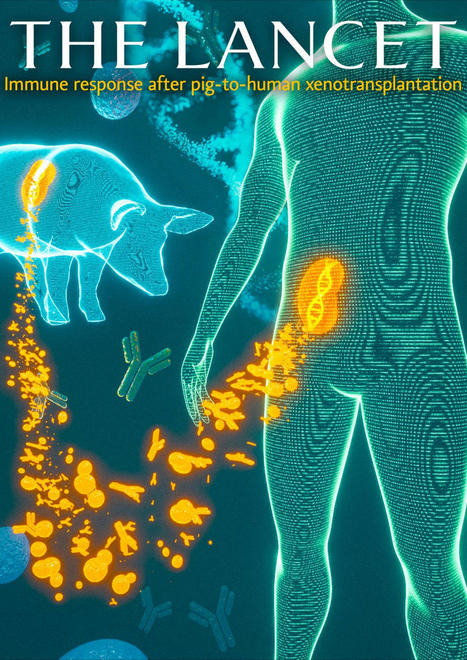
✨ Discovering New Horizons on #XENOTRANSPLANTATION ✨
We’re on an exhilarating journey to unravel the mysteries of xeno immune response. Our passion for… | 17 comments on LinkedIn

|
Scooped by Gilbert C FAURE |
The study unveils that potent combination therapies remodel TAMs to antitumor proinflammatory M1-like macrophages crucial for their therapeutic efficacy. I

|
Scooped by Gilbert C FAURE |
The future of vaccine development in light of accelerated technological advances through the COVID-19 pandemic.

|
Scooped by Gilbert C FAURE |
A description of H. Golding's research program and related publications.

|
Scooped by Gilbert C FAURE |
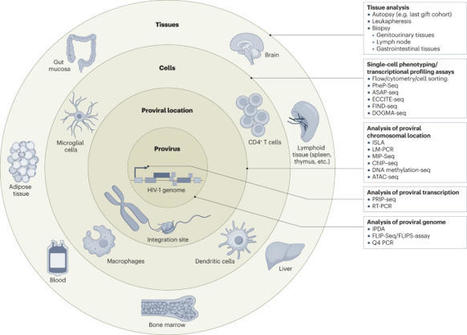
Successful approaches for eradication or cure of HIV-1 infection are likely to include immunological mechanisms, but remarkably little is known about how human immune responses can recognize and interact with the few HIV-1-infected cells that harbour genome-intact viral DNA, persist long term despite antiretroviral therapy and represent the main barrier to a cure. For a long time regarded as being completely shielded from host immune responses due to viral latency, these cells do, on closer examination with single-cell analytic techniques, display discrete footprints of immune selection, implying that human immune responses may be able to effectively engage and target at least some of these cells. The failure to eliminate rebound-competent virally infected cells in the majority of persons likely reflects the evolution of a highly selected pool of reservoir cells that are effectively camouflaged from immune recognition or rely on sophisticated approaches for resisting immune-mediated killing. Understanding the fine-tuned interplay between host immune responses and viral reservoir cells will help to design improved interventions that exploit the immunological vulnerabilities of HIV-1 reservoir cells. Finding a cure for HIV-1 infection, once considered elusive, now represents a major priority for the global microbiology research community. In this article, Armani-Tourret, Lichterfeld and colleagues highlight recent advances in understanding immunological vulnerabilities of virally infected cells that persist lifelong and represent the major barrier to a cure.

|
Rescooped by Gilbert C FAURE from Genetic Engineering Publications - GEG Tech top picks |
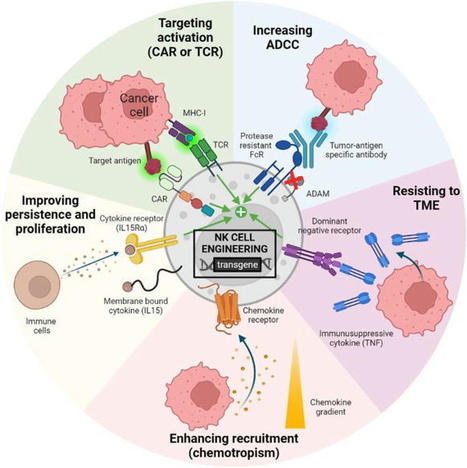
Review synthesizes research on NK cells' role in cancer immunity and their potential in therapeutics through bioengineering, immune checkpoint inhibitors, and cell engagers, highlighting ongoing preclinical and clinical trials.
Via BigField GEG Tech

In a recent study published in the journal Nature, researchers have compiled the available literature on natural killer (NK) cells, innate immune cells involved in the recognition and elimination of cells in distress, particularly virus-infected cells and tumors. They focus on reviewing current preclinical and clinical research in the field of NK therapies, primarily elucidating the role of NK cells in cancer immunity. They also explore the potential of bioengineering approaches to harness NK cells via the development of genetically modified NK cells, immune checkpoint inhibitors and cell engagement agents. The study reveals that, despite less than two decades of research in the field, NK cells are emerging as a safe, practical and potentially widely accessible means of clinical therapy, particularly antitumor. Although challenges exist in the adoption of NK cell therapies by conventional medicine, studies aimed at overcoming these challenges are already underway, bringing the future of clinical NK cell interventions closer than ever.

|
Scooped by Gilbert C FAURE |
Breakthrough sickle cell treatments are available in the U.S., but their high cost is forcing insurers to get creative.